|
(11) | EP 0 607 478 A1 |
| (12) | EUROPEAN PATENT APPLICATION |
|
|
|
|
||||||||||||||||||||||||
| (54) | Spectrally sensitized silver halide photographic materials |
| (57) The present invention refers to a silver halide photographic material containing
a support having coated thereon at least a silver halide emulsion layer spectrally
sensitized with a first sensitizing dye according to the formula (I):
wherein Z₁ and Z₂ each independently represents the atoms necessary to complete a substituted or unsubstituted 5-membered nitrogen-containing heterocyclic group; R and R₁ each independently represents a substituted or unsubstituted alkyl group; Q and Q₁ in combination represent the atoms required to complete a thiazolidinone nucleus; L₁, L₂ and L₃ each represents a methine group or a substituted methine group of the formula: wherein R₂ is an optionally substituted alkyl group having 1 to 6 carbon atoms or an optionally substituted aryl group having 6 to 10 carbon atoms; X⁻ is an anion and each of m, n and p is 0 or 1, provided that p is 0 when the dye forms an intramolecular salt; and a second sensitizing dye according to the formula (II): wherein Z₃ represents the atoms necessary to complete a 6-membered nitrogen-containing heterocyclic group; Z₄ represents the atoms necessary to complete a 5-membered nitrogen-containing heterocyclic group; R₃ and R₄ each represents a hydrogen atom, an alkyl group or a phenyl group; R, R₁, m, n, X and p have the same meaning of formula (I). The silver halide photographic material has the advantage to have a broad sensitivity in the red portion of the spectrum. It can be exposed to laser beam sources in the range of 600-700 nm. |
FIELD OF THE INVENTION
[0001] The present invention refers to a spectrally sensitized silver halide photographic material having broad sensitivity in the red portion of the spectrum.
BACKGROUND OF THE ART
[0002] Silver halide photography involves the exposure of silver halide with radiation to form a latent image that is developed during photographic processing to form a visible image. Silver halide is intrinsically sensitive only to light in the blue and ultraviolert regions of the spectrum. Thus, when silver halide is to be exposed to other wavelengths of radiation, such as green or red light in a multicolor element, a spectral sensitizing dye is required.
[0003] A Helium-neon laser is a typical light source emitting in the red portion of the spectrum (632.8 nm). The advent of solid state diodes that emit red radiation, having wavelengths in the range from 650 to 750 nm, has expanded the useful applications of red-sensitive photographic elements. Because of the wide variety of emission wavelengths, it would be desirable for a red-sensitive photographic material to have broad sensitivity in the red portion of the spectrum. This would allow a single material to be used with light sources, such as lasers and diodes, having a variety of emission wavelengths. Such broad sensitivity can generally be provided by either using a single sensitizing dye that provides broad sensitivity or by a combination of sensitizing dyes (usually two) that, by themselves, would provide narrower sensitivity.
[0004] British Patent No. 1,471,701 describes a silver halide photographic emulsion containing at least one trinuclear cyanine sensitizing dye having a 4-thiazolidinone nucleus. This emulsion is highly sensitive to light from a He-Ne laser light source (632.8 nm) and has high contrast. On the contrary, this emulsion is not sensitive to light from a laser diode emitting at higher wavelengths (around 670 nm).
[0005] Trinuclear cyanine sensitizing dyes containing a 2-quinolinyl residue have been described in US Patent No. 3,674,499 to obtain a supersensitized silver halide emulsion when combined with a condensate of formalin with a halogenated polyhydroxy benzene or a halogenated cresol, and in US Patent No. 3,877,943, to obtain a heat developable photographic material having high sensitivity and high density.
[0006] Many dye combinations have disadvantages, such as poor sensitivity, e.g. due to desensitization, or poor keeping stability, e.g. formation of fog during keeping.
[0007] It is an object of the present invention to provide silver halide photographic materials with broad sensitivity in the red portion of the spectrum without incurring the above-described problems.
SUMMARY OF THE INVENTION
[0008] The present invention refers to a silver halide photographic material containing a support having coated thereon at least a silver halide emulsion layer spectrally sensitized with a first sensitizing dye according to the formula (I):
wherein Z₁ and Z₂ each independently represents the atoms necessary to complete a substituted or unsubstituted 5-membered nitrogen-containing heterocyclic group; R and R₁ each independently represents a substituted or unsubstituted alkyl group; Q and Q₁ in combination represent the atoms required to complete a thiazolidinone nucleus; L₁, L₂ and L₃ each represents a methine group or a substituted methine group of the formula:
wherein R₂ is an optionally substituted alkyl group having 1 to 6 carbon atoms or an optionally substituted aryl group having 6 to 10 carbon atoms; X⁻ is an anion and each of m, n and p is 0 or 1, provided that p is 0 when the dye forms an intramolecular salt;
and a second sensitizing dye according to the formula (II):
wherein Z₃ represents the atoms necessary to complete a substituted or unsubstituted 6-membered nitrogen-containing heterocyclic group optionally containing substituent(s) and/or fused ring(s); Z₄ represents the atoms necessary to complete a substituted or unsubstituted 5-membered nitrogen-containing heterocyclic group optionally containing substituent(s) and/or fused ring(s); R₃ and R₄ each represents a hydrogen atom, an alkyl group or a phenyl group; R, R₁, m, n, X and p have the same meaning of formula (I).
[0009] The above described dye combination provides broad sensitivity in the red region of the spectrum with good speed, contrast and minimal residual color stain after processing.
DETAILED DESCRIPTION OF THE INVENTION
[0010] In the formula (I) of the first sensitizing dye of the present invention, Z₁ and Z₂ each independently represents the atoms necessary to complete a substituted or unsubstituted 5-membered nitrogen-containing heterocyclic nucleus such as thiazole, benzothiazole, naphthothiazole. These nuclei may be substituted by any of a number of groups known to be substituents for such nuclei. These includes sulfo, halogen (e.g. chloro, fluoro), alkyl of 1 to 12 carbon atoms (preferably of about 1 to 4 carbon atoms, e.g. methyl, ethyl, butyl, which may themselves be substituted with known elements such as hydroxy, halogen or sulfo), alkoxy of 1 to 12 carbon atoms (preferably of about 1 to 4 carbon atoms, e.g. methoxy, ethoxy, butoxy), carboxy, carboxylate of from 1 to 4 carbon atom (e.g. methyl ester, ethyl ester), sulfonamido or carbonamido. Examples of useful nuclei for Z₁ and Z₂ include a thiazole nucleus, e.g. thiazole, 4-methylthiazole, 4-phenylthiazole, 5-methylthiazole, 5-phenylthiazole, 4,5-dimethyl-thiazole, 4,5-diphenylthiazole, 4-(2-thienyl)-thiazole, benzothiazole, 4-chlorobenzothiazole, 5-chlorobenzothiazole, 6-chlorobenzothiazole, 7-chlorobenzothiazole, 4-methylbenzothiazole, 5-methylbenzothiazole, 6-methylbenzothiazole, 5-bromobenzothiazole, 6-bromobenzothiazole, 5-phenylbenzothiazole, 6-phenylbenzothiazole, 4-methoxybenzothiazole, 5-methoxybenzothiazole, 6-methoxybenzothiazole, 5-iodobenzothiazole, 6-iodobenzothiazole, 4-ethoxybenzothiazole, 5-ethoxybenzothiazole, tetrahydrobenzothiazole, 5,6-dimethoxybenzothiazole, 5,6-dioxymethylenebenzothiazole, 5-hydroxybenzothiazole, 6-hydroxybenzothiazole, naphtho(2,1-d)thiazole, naphtho(1,2-d)thiazole, 5-methoxynaphtho(2,3-d)thiazole, 5-ethoxynaphtho-(2,3-d)-thiazole, 8-methoxynaphtho(2,3-d)thiazole, 7-methoxynaphtho(2,3-d)thiazole, 4'-methoxythianaphtheno-7',6'-4,5-thiazole, etc.
[0011] R and R₁ each independently represents a substituted or unsubstituted alkyl of 1 to 20 carbon atoms (preferably of from 1 to 6 carbon atoms). Examples of alkyl include methyl, ethyl, propyl, isopropyl, butyl, octyl, etc. and substituted alkyl groups (preferably a substituted lower alkyl of from 1 to 6 carbon atoms), such as a hydroxyalkyl group, e.g., β-hydroxyethyl, γ-hydroxypropyl, δ-hydroxybutyl, etc., a carboxyalkyl group, e.g., β-carboxyethyl, γ-carboxypropyl, etc., a sulfoalkyl group, e.g., β-sulfoethyl, δ-sulfopropyl, γ-sulfobutyl, δ-sulfobutyl, etc., a sulfatoalkyl group, e.g., β-sulfatoethyl, γ-sulfatopropyl, etc., or an acyloxyalkyl group, e.g., β-acetoxyethyl, γ-acetoxypropyl, γ-propoxypropyl, etc.
[0012] Q and Q₁ in combination represent the atoms necessary to complete a thiazolidinone nucleus such as a 4-thiazolidinone nucleus or a 5-thiazolidinone nucleus and may be substituted as known to one skilled in the art. Examples of substituents include substituted or unsubstituted alkyl group of 1 to 12 carbon atoms such as methyl, ethyl or propyl, an allyl group, a substituted or ubsubstituted aralkyl group of 1 to 6 carbon atoms in the alkyl moiety thereof such as benzyl or p-carboxyphenylmethyl, an aryl group of 6 to 12 carbon atoms, e.g. a phenyl group or a p-carboxylphenyl, a hydroxyalkyl group, e.g. β-hydroxyethyl, a carboxyalkyl group, e.g. carboxymethyl, an alkoxycarbonylalkyl group, e.g. methoxycarbonylethyl, halogen (e.g chloro, fluoro), hydroxy, alkoxy (e.g. methoxy, ethoxy) and other conventional dyes substituents that would be apparent to one skilled in the art.
[0013] L₁, L₂ and L₃ each represents a methine group or a substituted methine group of the formula :
wherein R₂ may be unsubstituted alkyl having 1 to 6 carbon atoms, such as methyl or ethyl or a substituted alkyl group having 1 to 6 carbon atoms substituted with, for example an alkoxy group, a carboxy group, an alkoxycarbonyl group, or an aryl group and, more specifically, β-ethoxyethyl, β-carboxyethyl, β-methoxycarbonylethyl, benzyl or phenethyl, an unsubstituted aryl group or an aryl group substituted with an alkoxy group (e.g. having 1 to 6 carbon atoms), a halogen atom (e.g. fluorine, chlorine, bromine or iodine) or a carboxy group, for example phenyl, p-methoxyphenyl, p-chlorophenyl or o-carboxyphenyl.
[0014] The anion represented by X⁻, although not particularly restricted is, for example, a halogen ion (e.g., chloride, bromide, iodide), p-toluene sulfonate (PTS⁻), ethylsulfonate, perchlorate, or the like.
[0015] Each of m, n and p is 0 or 1, provided that p is 0 when the dye forms an intramolecular salt.
[0016] When the term "group" or "nucleus" is used in this invention to describe a chemical compound or substituent, the described chemical material includes the basic group or nucleus and that group or nucleus with conventional substitution. Where the term "moiety" is used to describe a chemical compound or substituent, only an unsubstituted chemical material is intended to be included. For example, "alkyl group" includes not only such alkyl moieties such as methyl, ethyl, octyl, stearyl, etc., but also such moieties bearing substituents groups such as halogen, cyano, hydroxyl, nitro, amine, carboxylate, etc. On the other hand, "alkyl moiety" or "alkyl" includes only methyl, ethyl, octyl, stearyl, cyclohexyl, etc.
[0017] Examples of the first sensitizing dyes according to this invention include the following. However, the scope of this invention is not limited to only these compounds.
The sensitizing dyes (I.1) to (I.9) of the present invention can be easily sensitized by one skilled in the art by reference to M.Hamer, "The Cyanine Dye and Related Compounds", J.Wiley & Sons Ltd. (1964), German Patents Nos. 971,941 and 1,013,167 or US Patent Reissue No. 29,476.
[0018] In the formula (II) of the present invention, Z₃ represents the atoms necessary to complete a substituted or unsubstituted 6-membered nitrogen-containing heterocyclic nucleus such as 2-quinoline group, 4-quinoline group and the like, optionally containing substituent(s) and/or fused ring(s). These nuclei may be substituted by any of a number of groups known to be substituents for such nuclei. These includes sulfo, halogen (e.g. chloro, fluoro), alkyl of 1 to 12 carbon atoms (preferably of about 1 to 4 carbon atoms, e.g. methyl, ethyl, butyl, which may themselves be substituted with known elements such as hydroxy, halogen or sulfo), alkoxy of 1 to 12 carbon atoms (preferably of about 1 to 4 carbon atoms, e.g. methoxy, ethoxy, butoxy), carboxy, carboxylate of from 1 to 4 carbon atom (e.g. methyl ester, ethyl ester), sulfonamido or carbonamido. Examples of useful nuclei for Z₃ include a 2-quinoline nucleus, e.g. quinoline, 3-methylquinoline, 5-methylquinoline, 6-chloroquinoline, a 4-quinoline nucleus, e.g. quinoline, 6-methoxyquinoline, 7-methylquinoline, 8-methylquinoline, and the like.
[0019] Z₄ represents heterocyclic rings which include all of those defined by the rings completed by Z₁ or Z₂ of the previous formula (I).
[0020] R₃ and R₄ each independently represents a hydrogen atom, an alkyl group of 1 to 18 carbon atoms (preferably of 1 to 12 carbon atoms such as methyl, ethyl, propyl, isopropyl, butyl, octyl, etc.) or a phenyl group (such as phenyl, carboxylphenyl).
[0021] Examples of the second sensitizing dyes according to this invention include the following exemplary compounds. However, the scope of this invention is not limited to only these compounds.
Synthesis of compound (II.1)
[0022] 3.79 g of 5-(1-ethylquinolin-2-yl)ethylidene-2-ethylthio-3-ethyl rhodaninium tosylate (0.007 mole), 2.79 g of 2-methyl-3-ethyl-naphthothiazolium tosylate (0.007 mole), 60 ml of ethanol and 6 ml of triethylamine were refluxed with stirring for 20 minutes. 1.44 g of NaBr (0.014 mole) were then added and the mixture was refluxed for other 10 minutes. The precipitates were collected by filtration and washed with methanol. The product was then boiled in acetone, filtered, washed with water and dried under vacuum. The product structure was confirmed by NMR.
Synthesis of compound (II.7)
[0023] 21.6 g of 5-(1-ethylquinolin-2-yl)ethylidene-2-ethylthio-3-ethyl rodaninium tosylate (0.039 mole), 16.0 g of 2-methyl-3-ethyl-naphthothiazolium tosylate (0.039 mole), 340 ml of ethanol and 34 ml of triethylamine were refluxed with stirring for 30 minutes. The mixture was left at room temperature overnight and then separated solid was filtered, then suspended in 500 ml of acetone, refluxed for 15 minutes, filtered and dried under vacuum. The product structure was confirmed by NMR.
[0024] The present invention also refers to a method for forming a silver image comprising image-wise exposing by means of a light source emitting light in the wavelength range of from 600 to 690 nm a photographic material containing a support having coated thereon at least a silver halide emulsion layer incorporating a first sensitizing dye and a second sensitizing dye spectrally sensitizing said emulsion for said wavelength range or for a substantial part of said wavelength range and wet-processing the exposed photographic material with aqueous developing or activating solutions and fixing solutions, thereby dissolving away said dyes from the resulting silver-image-containing material, wherein said first sensitizing dye is represented by the formula (I):
wherein Z₁ and Z₂ each independently represents the atoms necessary to complete a substituted or unsubstituted 5-membered nitrogen-containing heterocyclic group; R and R₁ each independently represents a substituted or unsubstituted alkyl group; Q and Q₁ in combination represent the atoms required to complete a thiazolidinone nucleus; L₁, L₂ and L₃ each represents a methine group or a substituted methine group of the formula
wherein R₂ is an optionally substituted alkyl group having 1 to 6 carbon atoms or an optionally substituted aryl group having 6 to 10 carbon atoms; X⁻ is an anion and each of m, n and p is 0 or 1, provided that p is 0 when the dye forms an intramolecular salt;
and wherein second sensitizing dye is represented by formula (II):
wherein Z₃ represents the atoms necessary to complete a substituted or unsubstituted 6-membered nitrogen-containing heterocyclic group optionally containing substituent(s) and/or fused ring(s); Z₄ represents the atoms necessary to complete a substituted or unsubstituted 5-membered nitrogen-containing heterocyclic group optionally containing substituent(s) and/or fused ring(s); R₃ and R₄ each represents a hydrogen atom, an alkyl group or a phenyl group; R, R₁, m, n, X and p have the same meaning of formula (I).
[0025] The first and second spectral sensitizing dyes of the present invention can be prepared according to well-known procedures in the art, such those described in James, The Theory of Phototgraphic Processes, MacMillan, 4th Edition, 1977.
[0026] The first and second spectral sensitizing dye of the present invention spectrally sensitize silver halide emulsions to radiation in the wavelength range of from 600 to 700 nm, especially from 620 to 680 nm, to provide photographic elements which are particularly suitable with a number of commercially available laser diodes. A further important advantage derived from the combined use of said first and said second spectral sensiziting dyes of the present invention is that the photographic material containing said sensitizing dyes exhibits an increased contrast and a reduced residual color stain after processing compared to the materials containing only one of the two spectral sensitizing dye.
[0027] The first spectral sensitizing dyes of the present invention are incorporated in the silver halide photographic emulsion in a content of from 0.01 to 0.20 grams/mol of silver, preferably from 0.02 to 0.16 grams/mol of silver.
[0028] The second spectral sensitizing dyes of the present invention are incorporated in the silver halide photographic emulsion in a content of from 0.005 to 0.20 grams/mol of silver, preferably from 0.01 to 0.15 grams/mol of silver.
[0029] The spectral sensitizing dyes of the present invention can be directly dispersed in the emulsion. Alternatively, they may be first dissolved in a suitable solvent such as methyl alcohol, ethyl alcohol, methyl cellosolve, acetone, water, pyridine, or a mixture thereof to add them to the emulsion as a solution. Processes for adding the sensitizing dyes to the photographic emulsion are described, for example, in US Pat. Nos. 3,469,987, 3,676,147, 3,822,135, 4,199,360, and in US Pat. Nos. 2,912,343, 3,342,605, 2,996,287 and 3,429,835. The aforesaid sensitizing dyes may be uniformly dispersed in the silver halide emulsion before coating on a suitable support. Of course, this dispersing procedure may be conducted in any suitable step of preparing the silver halide emulsion.
[0030] Any of the various types of photographic silver halide emulsions may be used in the practice of the present invention. Silver chloride, silver bromide, silver iodobromide, silver chlorobromide, silver chloroiodobromide, and mixtures thereof may be used, for example, dispersed in a hydrophilic colloid or carrier. Any configuration of grains, cubic, orthorombic, hexagonal, epitaxial, or tabular (high aspect ratio) grains may be used. The colloid may be partially hardened or fully hardened by any of the variously known photographic hardeners. Such hardeners are free aldehydes, aldehyde releasing compounds, triazines and diazines, aziridines, vinylsulfones, carbodiimides, and the like may be used, as described, for example, in US Pat. Nos. 3,232,764, 2,870,013, 3,819,608, 3,325,287, 3,992,366, 3,271,175 and 3,490,911.
[0031] The silver halide emulsions may be chemically sensitized using the usual sensitizing agents. Sulfur containing compounds, gold and noble metal compounds, polyoxylakylene compounds are particularly suitable. Methods for chemically sensitizing silver halide emulsions are described, for example, in Research Disclosure 17643, Section III, 1978.
[0032] Other conventional photographic addenda such as coating aids, antistatic agents, acutance dyes, antihalation dyes and layers, antifoggants, latent image stabilizers, supersentizers, antikinking agents, high intensity reciprocity failure reducers, plasticizers, matting agents, developing agents, color couplers, absorbing and scattering materials, which may be added to the silver halide emulsions are described in Research Disclosure 17643, 1978.
[0033] Gelatin is generally used as hydrophilic colloid for the silver halide photographic elements of the present invention. As hydrophilic colloids, gelatin derivatives, natural substances such as albumin, casein, agar-agar, alginic acid and the like, and hydrophilic polymers such as polyvinyl alcohol, polyvinylpyrrolidone, cellulose ethers, partially hydrolized polyvinyl acetate, and the like can be used in addition to or instead of gelatin. Further, gelatin can be partially substituted with polymer latexes obtained by emulsion polymerization of vinyl monomers, such as polyethylacrylate latexes, to improve the physical characteristics of the photographic layers.
[0034] Support base used in the silver halide photographic materials of this invention can be any of the conventionally used support bases, such as glass, cloth, metal, film including for example cellulose acetate, cellulose acetate-butyrate, cellulose nitrate, polyester, polyamine, polystyrene, and the like, paper including baryta, coated paper, resin-coated paper, and the like.
[0035] The silver halide emulsions according to the present invention may be used as photosensitive emulsions for various photographic materials, such as high surface sensitivity or high internal sensitivity negative emulsions, surface-fogged or unfogged direct-positive emulsions, print-out emulsions, reversal emulsions, emulsions for black-and-white materials, for color materials, radiographic materials, transfer color materials, and the like.
[0036] The photographic materials according to this invention may be processed to form a visible image upon association of the silver halides with an alkaline aqueous medium in the presence of a developing agent contained in the medium or in the material, as known in the art. In the case of color photographic materials, the processing comprises at least a color developing bath and, optionally, a prehardening bath, a neutralizing bath, a first (black and white) developing bath, etc. These and other baths which complete the photographic processing (e. g., bleaching, fixing, bleach-fixing, intensifying, stabilizing and washing baths) are well known in the art and are described for instance in Research Disclosure 17643, 1978.
EXAMPLE
[0038] A cubic monodispersed 0.32 µm silver chlorobromide (40 mole percent bromide) was prepared as in Example N. 2 of the European Patent application No. 423,538. The resulting emulsion was gold and sulfur sensitized, then split in several parts and each part was optical sensitized according to the following scheme :
| Emulsion Samples | First Optical Sensitizer | Second Optical Sensitizer |
| 1 (comp.) | I.1 | none |
| 2 (inv.) | I.1 | II.1 |
| 3 (inv.) | I.1 | II.7 |
| 4 (inv.) | I.1 | II8 |
| 5 (comp.) | I.1 | Compound A |
| 6 (comp.) | I.1 | Compound B |
| 7 (comp.) | I.1 | Compound C |
| 8 (comp.) | none | II.1 |
| 9 (comp.) | none | II.8 |
The resulting emulsion samples were coated onto a poly(ethylene terephthalate) support in amount of 4.1 g/sqm as silver. The gelatin content in thus formed emulsion layer was 2.6 g/sqm. A protective layer of gelatin (0.7 g/sqm) was superposed over the emulsion layer. The samples were exposed, in a first case, at 60 Ergs/square centimeter with a laser diode Toshiba Told 9215 emitting a laser beam at 670 nm and, in a second case, to a Helium-neon laser source emitting a laser beam at 632.8 nm. The samples were then sensitometrically examined. The speed has been measured in
, wherein E is expressed in lux.seconds at a density of 1.0 above Dmin. The contrast corresponds to the absolute value of the sensitometric curve slope measured at the densities of 0.07 and 0.17 above Dmin. The results are shown in the following Table 1, wherein Speed1 and Contrast1 refers, respectively, to the speed and contrast values obtained when the samples were exposed at 670 nm, and Speed2 and Contrast2 refers, respectively, to the speed and contrast values obtained when the samples were exposed at 632.8 nm.
Table 1
| Samples | Speed 1 670 nm | Contrast 1 670 nm | Speed 2 632.8 nm | Contrast 2 632.8 nm | Residual stain |
| 1 (comp.) | < 1.00 | 0.60 | 3.33 | 1.08 | light blue |
| 2 (inv.) | 2.95 | 1.19 | 3.26 | 1.25 | light blue |
| 3 (inv.) | 2.95 | 1.24 | 3.31 | 1.12 | light blue |
| 4 (inv.) | 2.85 | 1.29 | 3.10 | 1.25 | light blue |
| 5 (comp.) | < 1.00 | < 0.50 | 3.34 | 0.91 | deep blue |
| 6 (comp.) | < 1.00 | < 0.50 | 3.03 | 1.58 | light blue |
| 7 (comp.) | 3.40 | 0.91 | 3.31 | 0.78 | deep blue |
| 8 (comp.) | 2.80 | 1.43 | 2.57 | 1.25 | light pink |
| 9 (comp.) | 2.79 | 1.65 | 2.52 | 0.48 | light pink |
[0039] Table 1 shows that comparison sample 1, obtained using a sensitizing dye of formula (I), presented very low speed and contrast when exposed at 670 nm. Samples 2 to 4, obtained using a sensitizing dye of formula (I) combined with sensitizing dyes of formula (II), presented good speed, contrast and residual stain when exposed either to 632.8 nm or to 670 nm. In fact, acceptable values of speed and contrast are respectively of at least 2.85 and of 1.10; a light residual stain is acceptable. In addition, samples 2 to 4 surprisingly presented an increased contrast when exposed at 632.8 compared with the same contrast of comparison sample 1. Comparison samples 5 to 7, obtained using a sensitizing dye of formula (I) combined with a comparison sensitizing dye, presented bad speed when exposed at 670 nm (samples 5 and 6) , bad contrast (much less than the acceptable value of 1.10) and bad residual stain (deep blue is not acceptable). Further, comparison samples 8 and 9, obtained using a sensitizing dye of formula (II), presented bad speed when exposed at 632.8 nm.
1. Silver halide photographic material containing a support having coated thereon at
least a silver halide emulsion layer spectrally sensitized with a first sensitizing
dye according to the formula (I):
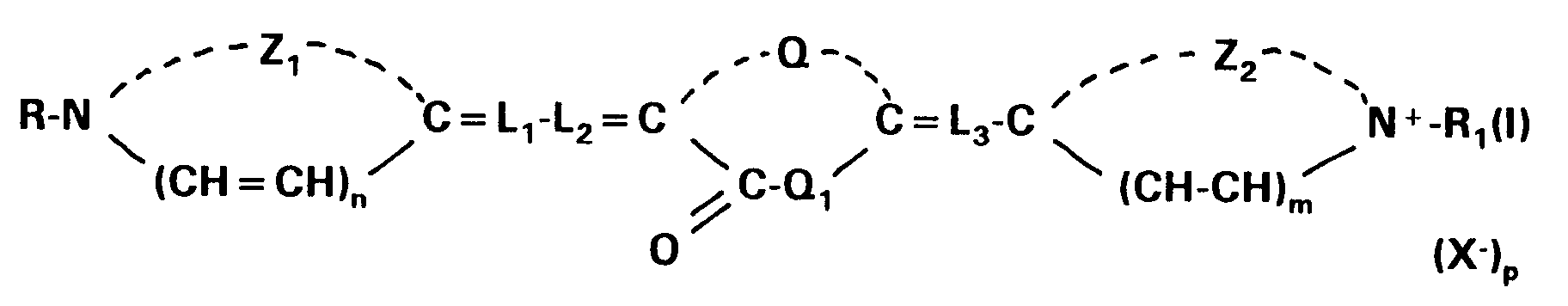
wherein Z₁ and Z₂ each independently represents the atoms necessary to complete a 5-membered nitrogen-containing heterocyclic group; R and R1 each independently represents an alkyl group; Q and Q1 in combination represent the atoms required to complete a thiazolidinone nucleus; L1, L2 and L3 each represents a methine group or a substituted methine group of the formula:
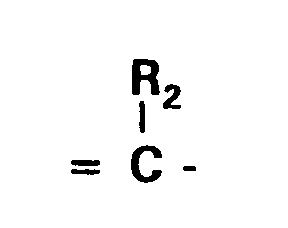
wherein R₂ is an alkyl group having 1 to 6 carbon atoms or an aryl group having 6 to 10 carbon atoms ; X⁻ is an anion and each of m, n and p is 0 or 1, provided that p is 0 when the dye forms an intramolecular salt;
and a second sensitizing dye according to the formula (II):
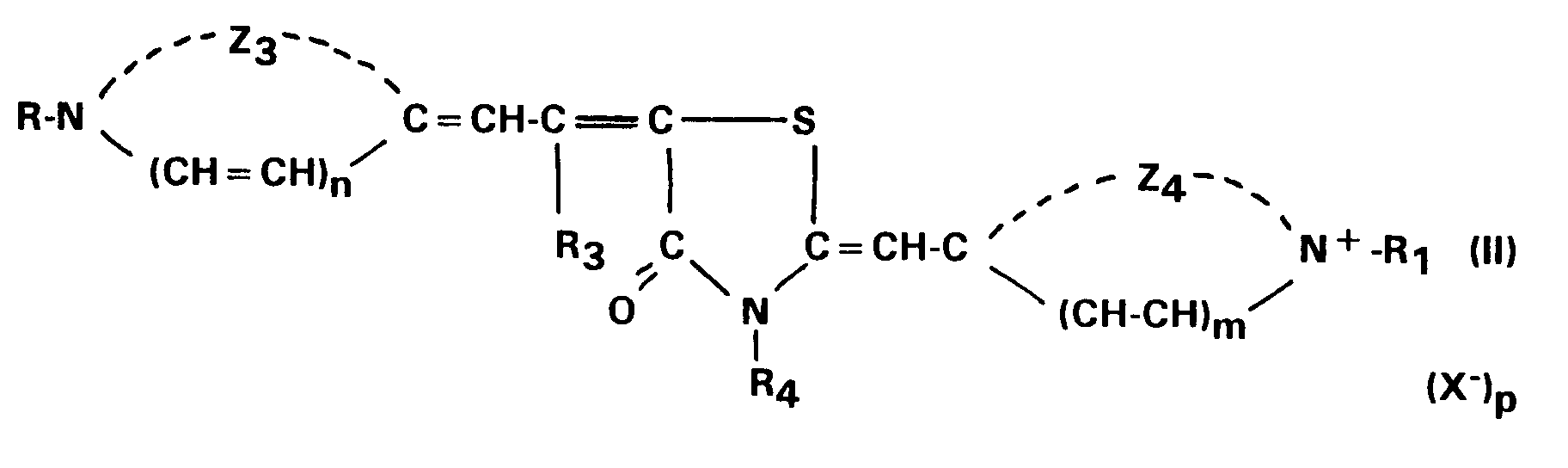
wherein Z₃ represents the atoms necessary to complete a 6-membered nitrogen-containing heterocyclic group; Z₄ represents the atoms necessary to complete a 5-membered nitrogen-containing heterocyclic group; R₃ and R₄ each represents a hydrogen atom, an alkyl group or a phenyl group; R, R₁, m, n, X and p have the same meaning of formula (I).
wherein Z₁ and Z₂ each independently represents the atoms necessary to complete a 5-membered nitrogen-containing heterocyclic group; R and R1 each independently represents an alkyl group; Q and Q1 in combination represent the atoms required to complete a thiazolidinone nucleus; L1, L2 and L3 each represents a methine group or a substituted methine group of the formula:
wherein R₂ is an alkyl group having 1 to 6 carbon atoms or an aryl group having 6 to 10 carbon atoms ; X⁻ is an anion and each of m, n and p is 0 or 1, provided that p is 0 when the dye forms an intramolecular salt;
and a second sensitizing dye according to the formula (II):
wherein Z₃ represents the atoms necessary to complete a 6-membered nitrogen-containing heterocyclic group; Z₄ represents the atoms necessary to complete a 5-membered nitrogen-containing heterocyclic group; R₃ and R₄ each represents a hydrogen atom, an alkyl group or a phenyl group; R, R₁, m, n, X and p have the same meaning of formula (I).
2. Silver halide photographic material as described in claim 1 wherein Z₁ and Z₂ each
independently represents a thiazole, a benzothiazole or a naphthothiazole nucleus.
3. Silver halide photographic material as described in claim 1 wherein Z₃ represents
a quinoline group.
4. Silver halide photographic material as described in claim 1 wherein Z₄ represents
a thiazole, a benzothiazole or a naphthothiazole nucleus.
5. Method for forming a silver image comprising image-wise exposing by means of a light
source emitting light in the wavelength range of from 600 to 690 nm a photographic
material containing a support having coated thereon at least a silver halide emulsion
layer incorporating a first sensitizing dye and a second sensitzing dye spectrally
sensitzing said emulsion for said wavelength range or for a substantial part of said
wavelength range and wet-processing the exposed photographic material with aqueous
developing or activating solutions and fixing solutions, thereby dissolving away said
dyes from the resulting silver-image-containing material, wherein said first sensitizing
dye is represented by the formula (I):
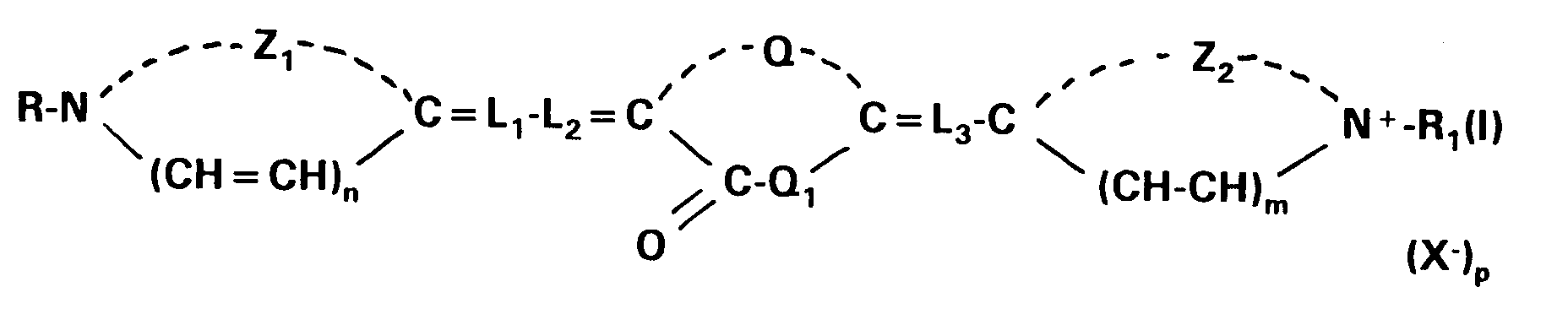
wherein Z₁ and Z₂ each independently represents the atoms necessary to complete a 5-membered nitrogen-containing heterocyclic group; R and R₁ each independently represents an alkyl group; Q and Q₁ in combination represent the atoms required to complete a thiazolidinone nucleus; L₁, L₂ and L₃ each represents a methine group or a substituted methine group of the formula:
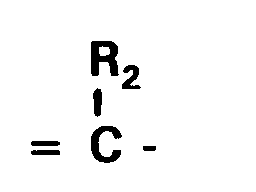
wherein R₂ is an alkyl group having 1 to 6 carbon atoms or an aryl group having 6 to 10 carbon atoms ; X⁻ is an anion and each of m, n and p is 0 or 1, provided that p is 0 when the dye forms an intramolecular salt;
and wherein second sensitizing dye is represented by formula (II):

wherein Z₃ represents the atoms necessary to complete a 6-membered nitrogen-containing heterocyclic group; Z₄ represents the atoms necessary to complete a 5-membered nitrogen-containing heterocyclic group; R₃ and R₄ each represents a hydrogen atom, an alkyl group or a phenyl group; R, R₁, m, n, X and p have the same meaning of formula (I).
wherein Z₁ and Z₂ each independently represents the atoms necessary to complete a 5-membered nitrogen-containing heterocyclic group; R and R₁ each independently represents an alkyl group; Q and Q₁ in combination represent the atoms required to complete a thiazolidinone nucleus; L₁, L₂ and L₃ each represents a methine group or a substituted methine group of the formula:
wherein R₂ is an alkyl group having 1 to 6 carbon atoms or an aryl group having 6 to 10 carbon atoms ; X⁻ is an anion and each of m, n and p is 0 or 1, provided that p is 0 when the dye forms an intramolecular salt;
and wherein second sensitizing dye is represented by formula (II):
wherein Z₃ represents the atoms necessary to complete a 6-membered nitrogen-containing heterocyclic group; Z₄ represents the atoms necessary to complete a 5-membered nitrogen-containing heterocyclic group; R₃ and R₄ each represents a hydrogen atom, an alkyl group or a phenyl group; R, R₁, m, n, X and p have the same meaning of formula (I).
