|
(11) | EP 2 574 656 B1 |
| (12) | EUROPEAN PATENT SPECIFICATION |
|
|
| (54) |
Lubricating oil compositions comprising p-alkoxy-N,N-dialkyl-aniline Schmierstoffzusammensetzung enthaltend p-Alkoxy-N,N-dialkyl-Anilin Composition lubrifiante contenant une p-alkoxy-N,N-dialkyl-aniline |
|
|
||||||||||||||||||||||||||||
| Note: Within nine months from the publication of the mention of the grant of the European patent, any person may give notice to the European Patent Office of opposition to the European patent granted. Notice of opposition shall be filed in a written reasoned statement. It shall not be deemed to have been filed until the opposition fee has been paid. (Art. 99(1) European Patent Convention). |
[0001] This invention relates to lubricating oil compositions including a class of aniline compounds. The aniline compounds are useful as ashless TBN (Total Base Number) boosters for lubricating oil compositions, particularly crankcase lubricating oil compositions having reduced levels of sulfated ash (SASH).
[0002] Environmental concerns have led to continued efforts to reduce the CO, hydrocarbon and nitrogen oxide (NOx) emissions from compression ignited (dieselfueled) and spark ignited (gasoline-fueled) light duty internal combustion engines. Further, there have been continued efforts to reduce the particulate emissions from compression ignited internal combustion engines. To meet the upcoming emission standards for heavy duty diesel vehicles, original equipment manufacturers (OEMs) will rely on the use of additional exhaust gas after-treatment devices. Such exhaust gas after-treatment devices may include catalytic converters, which may contain one or more oxidation catalysts, NOx storage catalysts, and/or NH3 reduction catalysts; and/or a particulate trap.
[0003] Oxidation catalysts can become poisoned and rendered less effective by exposure to certain elements/compounds present in engine exhaust gases, particularly by exposure to phosphorus and phosphorus compounds introduced into the exhaust gas by the degradation of phosphorus-containing lubricating oil additives. Reduction catalysts are sensitive to sulfur and sulfur compounds in the engine exhaust gas introduced by the degradation of both the base oil used to blend the lubricant, and sulfur-containing lubricating oil additives. Particulate traps can become blocked by metallic ash, which is a product of degraded metal-containing lubricating oil additives.
[0004] To ensure a long service life, lubricating oil additives that exert a minimum negative impact on such after-treatment devices must be identified, and OEM specifications for "new service fill" and "first fill" heavy duty diesel (HDD) lubricants require maximum sulfur levels of 0.4 mass %; maximum phosphorus levels of 0.12 mass %, and sulfated ash contents below 1.1 mass %, which lubricants are referred to as "mid-SAPS" lubricants (where "SAPS" is an acronym for "Sulfated Ash, Phosphorus, Sulfur"). In the future, OEMs may further restrict these levels maximum levels to 0.08 mass % phosphorus, 0.2 mass % sulfur and 0.8 mass % sulfated ash, with such lubricants being referred to as "low-SAPS" lubricating oil compositions.
[0005] As the amounts of phosphorus, sulfur and ash-containing lubricant additives are being reduced to provide mid- and low-SAPS lubricants that are compatible with exhaust gas after-treatment devices, the lubricating oil composition must continue to provide the high levels of lubricant performance, including adequate detergency, dictated by the "new service", and "first fill" specifications of the OEMs, such as the ACEA E6 and MB p228.51 (European) and API CI-4+ and API CJ-4 (U.S.) specifications for heavy duty engine lubricants. Criteria for being classified as a lubricating oil composition meeting the above listed industry standards are known to those skilled in the art.
[0006] The ability of a lubricant to neutralized acidic byproducts of combustion, which increases in engines provided with exhaust gas recirculation (EGR) systems, particularly condensed EGR systems in which exhaust gasses are cooled prior to recirculation, can be improved, and the drain interval of the lubricant can be extended, by increasing the total base number (TBN) of the composition. Historically, TBN has been provided by overbased detergents that introduce sulfated ash into the composition. It would be advantageous to provide a lubricating oil composition with a high level of TBN using a TBN boosting component that does not contribute sulfated ash. As highly basic components are known to induce corrosion and, in some cases reduce the compatibility between lubricating oil compositions and the fluoroelastomeric seal materials used in engines, it would be preferable to provide such a component that does not induce corrosion and, preferably, does not adversely affect seals compatibility. Due to demands for improved fuel economy, less viscous lubricants, such as 0W and 5W 20 and 30 grade lubricants have become more prevalent. To allow for easier formulation of such lubricants, the amount of polymer introduced by additives is preferably minimized. Therefore, it would be further preferable to provide a non-polymeric ashless TBN source.
[0007] US Patent Nos. 5,525,247; 5,672,570; and 6,569,818 are directed to "low ash" lubricating oil compositions in which sulfated ash content is reduced by replacing overbased detergents with neutral detergents. These patents describe such lubricants as providing sufficient detergency, but make no claim that such lubricants will provide sufficient TBN for use, for example, in HDD engines. US Patent Application 2007/0203031 describes the use of a high TBN nitrogen-containing dispersants as ashless TBN sources.
[0008] US Patent Nos. 4,100,082; 4,200,545; 4,320,021, 4,663,063; 4,708.809; and Russian Patent Application SU1825780 describe amino-phenol compounds as lubricating oil additives (e.g., dispersant/detergents). US Patent Nos. 2,511,750; 3,634,248; 4,269,720; 4,335,006; 4,411,805; and 6,242,394 describe certain aniline compounds as stabilizers (antioxidants) for lubricating oil compositions. US Patent No. 4,778,654 describes alkylaniline/formaldehyde co-oligomers useful as corrosion inhibitors.
[0009] The present Applicant's co-pending EP 2 206 764 A1 describes aniline compounds as ashless TBN sources for lubricating oils. A preference is expressed for di-alkyl substituted aniline compounds where both of the alkyl groups attached to the nitrogen atom are linear. An example of a di-branched (di-2-ethylhexyl) compound is provided but there are no data given for its TBN performance in a lubricating oil.
[0010] It has now been found that certain alkoxy-substituted di-alkyl aniline species are able to provide improved TBN performance when used as additives for lubricating oils. Such oil also have enhanched fluoroelastomer seal compatibility, as measured for example by the VW-6/AK6 test.
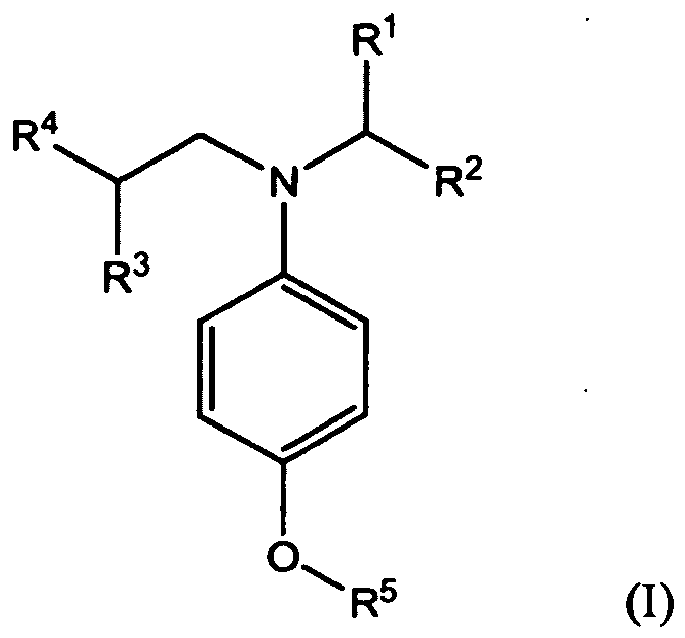
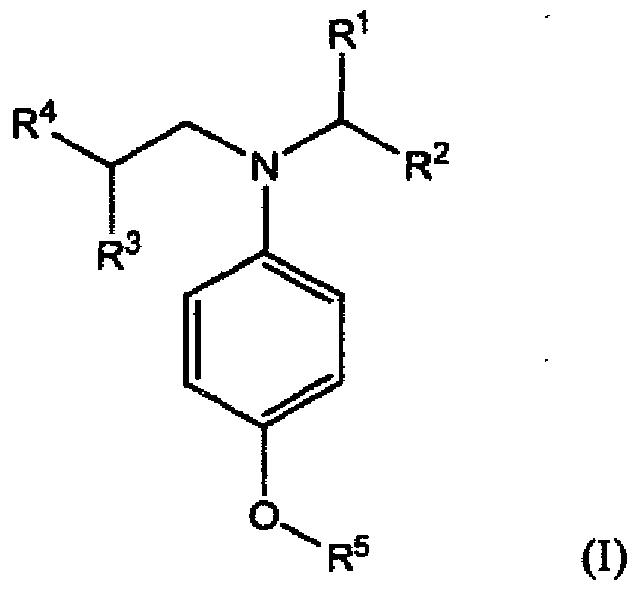
[0011] Accordingly in a first aspect, the present invention provides a lubricating oil composition comprising a major amount of oil of lubricating viscosity and a minor amount of one or more compounds of formula (I):
wherein R1, R2, R3, R4 and R5 independently represent alkyl or substituted alkyl having no aryl substituent.
[0012] The compounds of formula (I) are characterised in that they have two different branched alkyl groups attached to the nitrogen atom. One of the branched alkyl groups has a branch at a carbon atom in an α-position to the nitrogen atom and the other has a branch at a carbon atom in a β-position to the nitrogen atom. It has been found that this specific configuration provides enhanced TBN performance.
[0013] Preferably, the lubricating oils meet the performance criteria of one or more of the ACEA E6, MB p228.51, API CI-4+ and API CJ-4 specifications for heavy duty engine lubricants.
[0014] In accordance with a second aspect, the present invention provides a concentrate for the preparation of a lubricating oil composition according to any preceding claim, the concentrate comprising from about 2.5 to about 30 mass % of one or more compounds of formula (I); about 10 to about 40 mass % of a nitrogen-containing dispersant; about 2 to about 20 mass % of an aminic antioxidant, a phenolic antioxidant, a molybdenum compound, or a mixture thereof; about 5 to 40 mass % of a detergent; and from about 2 to about 20 mass % of a metal dihydrocarbyl dithiophosphate.
[0015] In accordance with a third aspect of the present invention, there is provided a compound of formula (I):
wherein R1, R2, R3, R4 and R5 independently represent alkyl or substituted alkyl having no aryl substituent.
[0016] In accordance with a fourth aspect of the present invention, there is provided a method of increasing the TBN of a lubricating oil composition without concurrently increasing the SASH content, the method comprising adding to the lubricating oil composition one or more compounds according to the third aspect.
[0017] In accordance with a fifth aspect, the present invention provides the use of a compound according to the third aspect as an ashless lubricating oil composition TBN source.
[0018] The compounds useful as ashless TBN sources for lubricating oil compositions have the formula (I):
[0022] Embodiments where R1 and/or R3 represent longer alkyl groups, for example alkyl groups having 3 to 12 carbon atoms are also within the scope of the present invention.
[0023] Preferably, R2 represents an alkyl group having 3 to 12 carbon atoms, which group may be linear or branched.
[0024] Preferably, R4 represents an alkyl group having 3 to 12 carbon atoms, which group may be linear or branched.
[0025] Preferably, R5 represents methyl or ethyl such that the benzene ring is methoxy or ethoxy substituted. More preferably, R5 represents ethyl. Embodiments where R5 represents a longer alkyl group, for example an alkyl group having 3 to 12 carbon atoms are also within the scope of the present invention.
[0028] Preferably, the compounds of the present invention have a TBN (as measured in accordance with ASTM D-2896 and/or ASTM D-4793) of at least about 50, preferably at least about 100, more preferably at least about 140, and most preferably at least about 180 mg KOH/g.
[0029] Preferably, compounds of the present invention have a >99% weight loss, as determined by thermal gravity analysis (at 10 °C/min temperature ramp rate in air) at a temperature of at least about 200, preferably at least about 250, more preferably at least about 300 °C.
[0030] Methods for forming compounds of formula (I) should be apparent to those skilled in the art. Some suitable methods are described below.
[0031] Aniline and substituted anilines such as ethoxyaniline are commercially available. N, N-dialkylaniline and substituted derivatives such as N,N-dialkylethoxyaniline can be prepared by reacting the appropriate aniline or substituted derivatives such as ethoxyaniline with aldehydes/ketones in a 1:2 or excess molar ratio in the presence of a reducing agent such as sodium triacetoxyborohydride in dichloromethane solvent. Such methods are well known and described in the literature, see for example Jn. Org. Chem (1996), 61, pp. 3489-3862.
[0032] Another method involves reacting the appropriate aniline or substituted aniline with halogenated alkyl (e.g., brominated alkyl) in a 2:1 molar ratio, in the presence triethylamine, in an acetonitrile solvent.
[0033] Yet another method reacts aniline or substituted derivatives such as ethoxyaniline with aldehydes/ketones in a 1:2 or excess molar ratio in the presence of hydrogen and 10% Pd/C catalyst in methanol solvent. Such methods are well known and a number of such methods are described, for example, in US Patent No. 2,045,574.)
[0034] The unsymmetrical compounds of the present invention can be made in analogous manners using the methods described above and others by employing a step-wise approach whereby one alkyl group is appended to the nitrogen atom before a second different alkyl group is attached.
[0035] Oils of lubricating viscosity useful in the context of the present invention may be selected from natural lubricating oils, synthetic lubricating oils and mixtures thereof. The lubricating oil may range in viscosity from light distillate mineral oils to heavy lubricating oils such as gasoline engine oils, mineral lubricating oils and heavy duty diesel oils. Generally, the viscosity of the oil ranges from about 2 centistokes to about 40 centistokes, especially from about 4 centistokes to about 20 centistokes, as measured at 100°C.
[0036] Natural oils include animal oils and vegetable oils (e.g., castor oil, lard oil); liquid petroleum oils and hydrorefined, solvent-treated or acid-treated mineral oils of the paraffinic, naphthenic and mixed paraffinic-naphthenic types. Oils of lubricating viscosity derived from coal or shale also serve as useful base oils.
[0037] Synthetic lubricating oils include hydrocarbon oils and halo-substituted hydrocarbon oils such as polymerized and interpolymerized olefins (e.g., polybutylenes, polypropylenes, propylene-isobutylene copolymers, chlorinated polybutylenes, poly(1-hexenes), poly(1-octenes), poly(1-decenes)); alkylbenzenes (e.g., dodecylbenzenes, tetradecylbenzenes, dinonylbenzenes, di(2-ethylhexyl)benzenes); polyphenyls (e.g., biphenyls, terphenyls, alkylated polyphenols); and alkylated diphenyl ethers and alkylated diphenyl sulfides and derivative, analogs and homologs thereof. Also useful are synthetic oils derived from a gas to liquid process from Fischer-Tropsch synthesized hydrocarbons, which are commonly referred to as gas to liquid, or "GTL" base oils.
[0038] Alkylene oxide polymers and interpolymers and derivatives thereof where the terminal hydroxyl groups have been modified by esterification, etherification, etc., constitute another class of known synthetic lubricating oils. These are exemplified by polyoxyalkylene polymers prepared by polymerization of ethylene oxide or propylene oxide, and the alkyl and aryl ethers of polyoxyalkylene polymers (e.g., methylpolyiso-propylene glycol ether having a molecular weight of 1000 or diphenyl ether of poly-ethylene glycol having a molecular weight of 1000 to 1500); and mono- and polycarboxylic esters thereof, for example, the acetic acid esters, mixed C3-C8 fatty acid esters and C13 oxo acid diester of tetraethylene glycol.
[0039] Another suitable class of synthetic lubricating oils comprises the esters of dicarboxylic acids (e.g., phthalic acid, succinic acid, alkyl succinic acids and alkenyl succinic acids, maleic acid, azelaic acid, suberic acid, sebasic acid, fumaric acid, adipic acid, linoleic acid dimer, malonic acid, alkylmalonic acids, alkenyl malonic acids) with a variety of alcohols (e.g., butyl alcohol, hexyl alcohol, dodecyl alcohol, 2-ethylhexyl alcohol, ethylene glycol, diethylene glycol monoether, propylene glycol). Specific examples of such esters includes dibutyl adipate, di(2-ethylhexyl) sebacate, di-n-hexyl fumarate, dioctyl sebacate, diisooctyl azelate, diisodecyl azelate, dioctyl phthalate, didecyl phthalate, dieicosyl sebacate, the 2-ethylhexyl diester of linoleic acid dimer, and the complex ester formed by reacting one mole of sebacic acid with two moles of tetraethylene glycol and two moles of 2-ethylhexanoic acid.
[0040] Esters useful as synthetic oils also include those made from C5 to C12 monocarboxylic acids and polyols and polyol esters such as neopentyl glycol, trimethylolpropane, pentaerythritol, dipentaerythritol and tripentaerythritol.
[0041] Silicon-based oils such as the polyalkyl-, polyaryl-, polyalkoxy- or polyaryloxysilicone oils and silicate oils comprise another useful class of synthetic lubricants; such oils include tetraethyl silicate, tetraisopropyl silicate, tetra-(2-ethylhexyl)silicate, tetra-(4-methyl-2-ethylhexyl)silicate, tetra-(p-tert-butyl-phenyl) silicate, hexa-(4-methyl-2-ethylhexyl)disiloxane, poly(methyl)siloxanes and poly(methylphenyl)siloxanes. Other synthetic lubricating oils include liquid esters of phosphorous-containing acids (e.g., tricresyl phosphate, trioctyl phosphate, diethyl ester of decylphosphonic acid) and polymeric tetrahydrofurans.
[0042] The oil of lubricating viscosity may comprise a Group I, Group II or Group III, base stock or base oil blends of the aforementioned base stocks. Preferably, the oil of lubricating viscosity is a Group II or Group III base stock, or a mixture thereof, or a mixture of a Group I base stock and one or more a Group II and Group III. Preferably, a major amount of the oil of lubricating viscosity is a Group II, Group III, Group IV or Group V base stock, or a mixture thereof. The base stock, or base stock blend preferably has a saturate content of at least 65%, more preferably at least 75%, such as at least 85%. Most preferably, the base stock, or base stock blend, has a saturate content of greater than 90%. Preferably, the oil or oil blend will have a sulfur content of less than 1%, preferably less than 0.6%, most preferably less than 0.4%, by weight.
[0043] Preferably the volatility of the oil or oil blend, as measured by the Noack volatility test (ASTM D5880), is less than or equal to 30%, preferably less than or equal to 25%, more preferably less than or equal to 20%, most preferably less than or equal 16%. Preferably, the viscosity index (VI) of the oil or oil blend is at least 85, preferably at least 100, most preferably from about 105 to 140.
[0044] Definitions for the base stocks and base oils in this invention are the same as those found in the American Petroleum Institute (API) publication "Engine Oil Licensing and Certification System", Industry Services Department, Fourteenth Edition, December 1996, Addendum 1, December 1998. Said publication categorizes base stocks as follows:
- a) Group I base stocks contain less than 90 percent saturates and/or greater than 0.03 percent sulfur and have a viscosity index greater than or equal to 80 and less than 120 using the test methods specified in Table 1.
- b) Group II base stocks contain greater than or equal to 90 percent saturates and less than or equal to 0.03 percent sulfur and have a viscosity index greater than or equal to 80 and less than 120 using the test methods specified in Table 1.
- c) Group III base stocks contain greater than or equal to 90 percent saturates and less than or equal to 0.03 percent sulfur and have a viscosity index greater than or equal to 120 using the test methods specified in Table 1.
- d) Group IV base stocks are polyalphaolefins (PAO).
- e) Group V base stocks include all other base stocks not included in Group I, II, III, or IV.
Table I - Analytical Methods for Base Stock
| Property | Test Method |
| Saturates | ASTM D 2007 |
| Viscosity Index | ASTM D 2270 |
| Sulfur | ASTM D 2622 |
| ASTM D 4294 | |
| ASTM D 4927 | |
| ASTM D 3120 |
[0045] Lubricating oil compositions of the present invention may further contain one or more metal-containing or ash-forming detergents. Metal-containing or ash-forming detergents function both as detergents to reduce or remove deposits and as acid neutralizers or rust inhibitors, thereby reducing wear and corrosion and extending engine life. Detergents generally comprise a polar head with a long hydrophobic tail, with the polar head comprising a metal salt of an acidic organic compound. The salts may contain a substantially stoichiometric amount of the metal in which case they are usually described as normal or neutral salts, and would typically have a total base number or TBN (as can be measured by ASTM D-2896 and/or ASTM D-4793) of from 0 to 80. A large amount of a metal base may be incorporated by reacting excess metal compound (e.g., an oxide or hydroxide) with an acidic gas (e.g., carbon dioxide). The resulting overbased detergent comprises neutralized detergent as the outer layer of a metal base (e.g. carbonate) micelle. Such overbased detergents may have a TBN of 150 or greater, and typically will have a TBN of from 250 to 450 or more. In the presence of the compounds of formula (I), the amount of overbased detergent can be reduced, or detergents having reduced levels of overbasing (e.g., detergents having a TBN of 100 to 200), or neutral detergents can be employed, resulting in a corresponding reduction in the SASH content of the lubricating oil composition without a reduction in the performance thereof.
[0046] Detergents that may be used include oil-soluble neutral and overbased sulfonates, phenates, sulfurized phenates, thiophosphonates, salicylates, and naphthenates and other oil-soluble carboxylates of a metal, particularly the alkali or alkaline earth metals, e.g., sodium, potassium, lithium, calcium, and magnesium. The most commonly used metals are calcium and magnesium, which may both be present in detergents used in a lubricant, and mixtures of calcium and/or magnesium with sodium. Particularly convenient metal detergents are neutral and overbased calcium sulfonates having TBN of from 20 to 450 TBN, and neutral and overbased calcium phenates and sulfurized phenates having TBN of from 50 to 450. Combinations of detergents, whether overbased or neutral or both, may be used.
[0047] Sulfonates may be prepared from sulfonic acids which are typically obtained by the sulfonation of alkyl substituted aromatic hydrocarbons such as those obtained from the fractionation of petroleum or by the alkylation of aromatic hydrocarbons. Examples included those obtained by alkylating benzene, toluene, xylene, naphthalene, diphenyl or their halogen derivatives such as chlorobenzene, chlorotoluene and chloronaphthalene. The alkylation may be carried out in the presence of a catalyst with alkylating agents having from about 3 to more than 70 carbon atoms. The alkaryl sulfonates usually contain from about 9 to about 80 or more carbon atoms, preferably from about 16 to about 60 carbon atoms per alkyl substituted aromatic moiety.
[0048] The oil soluble sulfonates or alkaryl sulfonic acids may be neutralized with oxides, hydroxides, alkoxides, carbonates, carboxylate, sulfides, hydrosulfides, nitrates, borates and ethers of the metal. The amount of metal compound is chosen having regard to the desired TBN of the final product but typically ranges from about 100 to 220 mass % (preferably at least 125 mass %) of that stoichiometrically required.
[0049] Metal salts of phenols and sulfurized phenols are prepared by reaction with an appropriate metal compound such as an oxide or hydroxide and neutral or overbased products may be obtained by methods well known in the art. Sulfurized phenols may be prepared by reacting a phenol with sulfur or a sulfur containing compound such as hydrogen sulfide, sulfur monohalide or sulfur dihalide, to form products which are generally mixtures of compounds in which 2 or more phenols are bridged by sulfur containing bridges.
[0050] Lubricating oil compositions of the present invention may further contain one or more ashless dispersants, which effectively reduce formation of deposits upon use in gasoline and diesel engines, when added to lubricating oils. Ashless dispersants useful in the compositions of the present invention comprises an oil soluble polymeric long chain backbone having functional groups capable of associating with particles to be dispersed. Typically, such dispersants comprise amine, alcohol, amide or ester polar moieties attached to the polymer backbone, often via a bridging group. The ashless dispersant may be, for example, selected from oil soluble salts, esters, aminoesters, amides, imides and oxazolines of long chain hydrocarbon-substituted mono-and polycarboxylic acids or anhydrides thereof; thiocarboxylate derivatives of long chain hydrocarbons; long chain aliphatic hydrocarbons having polyamine moieties attached directly thereto; and Mannich condensation products formed by condensing a long chain substituted phenol with formaldehyde and polyalkylene polyamine. The most common dispersant in use is the well known succinimide dispersant, which is a condensation product of a hydrocarbyl-substituted succinic anhydride and a poly(alkyleneamine). Both mono-succinimide and bis-succinimide dispersants (and mixtures thereof) are well known.
[0051] Preferably, the ashless dispersant is a "high molecular weight" dispersant having a number average molecular weight (Mn) greater than or equal to 4,000, such as between 4,000 and 20,000. The precise molecular weight ranges will depend on the type of polymer used to form the dispersant, the number of functional groups present, and the type of polar functional group employed. For example, for a polyisobutylene derivatized dispersant, a high molecular weight dispersant is one formed with a polymer backbone having a number average molecular weight of from about 1680 to about 5600. Typical commercially available polyisobutylene-based dispersants contain polyisobutylene polymers having a number average molecular weight ranging from about 900 to about 2300, functionalized by maleic anhydride (MW = 98), and derivatized with polyamines having a molecular weight of from about 100 to about 350. Polymers of lower molecular weight may also be used to form high molecular weight dispersants by incorporating multiple polymer chains into the dispersant, which can be accomplished using methods that are know in the art.
[0052] Preferred groups of dispersant include polyamine-derivatized poly α-olefin, dispersants, particularly ethylene/butene alpha-olefin and polyisobutylene-based dispersants. Particularly preferred are ashless dispersants derived from polyisobutylene substituted with succinic anhydride groups and reacted with polyethylene amines, e.g. polyethylene diamine, tetraethylene pentamine; or a polyoxyalkylene polyamine, e.g. polyoxypropylene diamine, trimethylolaminomethane; a hydroxy compound, e.g. pentaerythritol; and combinations thereof. One particularly preferred dispersant combination is a combination of (A) polyisobutylene substituted with succinic anhydride groups and reacted with (B) a hydroxy compound, e.g. pentaerythritol; (C) a polyoxyalkylene polyamine, e.g. polyoxypropylene diamine, or (D) a polyalkylene diamine, e.g. polyethylene diamine and tetraethylene pentamine using about 0.3 to about 2 moles of (B), (C) and/or (D) per mole of (A). Another preferred dispersant combination comprises a combination of (A) polyisobutenyl succinic anhydride with (B) a polyalkylene polyamine, e.g. tetraethylene pentamine, and (C) a polyhydric alcohol or polyhydroxy-substituted aliphatic primary amine, e.g. pentaerythritol or trismethylolaminomethane, as described in U.S. Patent No. 3,632,511.
[0053] Another class of ashless dispersants comprises Mannich base condensation products. Generally, these products are prepared by condensing about one mole of an alkyl-substituted mono- or polyhydroxy benzene with about 1 to 2.5 moles of carbonyl compound(s) (e.g., formaldehyde and paraformaldehyde) and about 0.5 to 2 moles of polyalkylene polyamine, as disclosed, for example, in U.S. Patent No. 3,442,808. Such Mannich base condensation products may include a polymer product of a metallocene catalyzed polymerization as a substituent on the benzene group, or may be reacted with a compound containing such a polymer substituted on a succinic anhydride in a manner similar to that described in U.S. Patent No. 3,442,808. Examples of functionalized and/or derivatized olefin polymers synthesized using metallocene catalyst systems are described in the publications identified supra.
[0054] The dispersant can be further post treated by a variety of conventional post treatments such as boration, as generally taught in U.S. Patent Nos. 3,087,936 and 3,254,025. Boration of the dispersant is readily accomplished by treating an acyl nitrogen-containing dispersant with a boron compound such as boron oxide, boron halide boron acids, and esters of boron acids, in an amount sufficient to provide from about 0.1 to about 20 atomic proportions of boron for each mole of acylated nitrogen composition. Useful dispersants contain from about 0.05 to about 2.0 mass %, e.g., from about 0.05 to about 0.7 mass % boron. The boron, which appears in the product as dehydrated boric acid polymers (primarily (HBO2)3), is believed to attach to the dispersant imides and diimides as amine salts, e.g., the metaborate salt of the diimide. Boration can be carried out by adding from about 0.5 to 4 mass %, e.g., from about 1 to about 3 mass % (based on the mass of acyl nitrogen compound) of a boron compound, preferably boric acid, usually as a slurry, to the acyl nitrogen compound and heating with stirring at from about 135°C to about 190°C, e.g., 140°C to 170°C, for from about 1 to about 5 hours, followed by nitrogen stripping. Alternatively, the boron treatment can be conducted by adding boric acid to a hot reaction mixture of the dicarboxylic acid material and amine, while removing water. Other post reaction processes commonly known in the art can also be applied.
[0055] The dispersant may also be further post treated by reaction with a so-called "capping agent". Conventionally, nitrogen-containing dispersants have been "capped" to reduce the adverse effect such dispersants have on the fluoroelastomer engine seals. Numerous capping agents and methods are known. Of the known "capping agents", those that convert basic dispersant amino groups to non-basic moieties (e.g., amido or imido groups) are most suitable. The reaction of a nitrogen-containing dispersant and alkyl acetoacetate (e.g., ethyl acetoacetate (EAA)) is described, for example, in U.S. Patent Nos. 4,839,071; 4,839,072 and 4,579,675. The reaction of a nitrogen-containing dispersant and formic acid is described, for example, in U.S. Patent No. 3,185,704. The reaction product of a nitrogen-containing dispersant and other suitable capping agents are described in U.S. Patent Nos. 4,663,064 (glycolic acid); 4,612,132; 5,334,321; 5,356,552; 5,716,912; 5,849,676; 5,861,363 (alkyl and alkylene carbonates, e.g., ethylene carbonate); 5,328,622 (mono-epoxide); 5,026,495; 5,085,788; 5,259,906; 5,407,591 (poly (e.g., bis)-epoxides) and 4,686,054 (maleic anhydride or succinic anhydride). The foregoing list is not exhaustive and other methods of capping nitrogen-containing dispersants are known to those skilled in the art.
[0056] For adequate piston deposit control, a nitrogen-containing dispersant can be added in an amount providing the lubricating oil composition with from about 0.03 mass % to about 0.15 mass %, preferably from about 0.07 to about 0.12 mass %, of nitrogen.
[0057] Ashless dispersants are basic in nature and therefore have a TBN which, depending on the nature of the polar group and whether or not the dispersant is borated or treated with a capping agent, may be from about 5 to about 200 mg KOH/g. However, high levels of basic dispersant nitrogen are known to have a deleterious effect on the fluoroelastomeric materials conventionally used to form engine seals and, therefore, it is preferable to use the minimum amount of dispersant necessary to provide piston deposit control, and to use substantially no dispersant, or preferably no dispersant, having a TBN of greater than 5. Preferably, the amount of dispersant employed will contribute no more than 4, preferably no more than 3 mg KOH/g of TBN to the lubricating oil composition. It is further preferable that dispersant provides no greater than 30, preferably no greater than 25% of the TBN of the lubricating oil composition.
[0058] Additional additives may be incorporated in the compositions of the invention to enable them to meet particular requirements. Examples of additives which may be included in the lubricating oil compositions are metal rust inhibitors, viscosity index improvers, corrosion inhibitors, oxidation inhibitors, friction modifiers, other dispersants, anti-foaming agents, anti-wear agents and pour point depressants. Some are discussed in further detail below.
[0059] Dihydrocarbyl dithiophosphate metal salts are frequently used as antiwear and antioxidant agents. The metal may be an alkali or alkaline earth metal, or aluminum, lead, tin, molybdenum, manganese, nickel or copper. The zinc salts are most commonly used in lubricating oil in amounts of 0.1 to 10, preferably 0.2 to 2 wt. %, based upon the total weight of the lubricating oil composition. They may be prepared in accordance with known techniques by first forming a dihydrocarbyl dithiophosphoric acid (DDPA), usually by reaction of one or more alcohol or a phenol with P2S5 and then neutralizing the formed DDPA with a zinc compound. For example, a dithiophosphoric acid may be made by reacting mixtures of primary and secondary alcohols. Alternatively, multiple dithiophosphoric acids can be prepared where the hydrocarbyl groups on one are entirely secondary in character and the hydrocarbyl groups on the others are entirely primary in character. To make the zinc salt, any basic or neutral zinc compound could be used but the oxides, hydroxides and carbonates are most generally employed. Commercial additives frequently contain an excess of zinc due to the use of an excess of the basic zinc compound in the neutralization reaction.
[0060] The preferred zinc dihydrocarbyl dithiophosphates are oil soluble salts of dihydrocarbyl dithiophosphoric acids and may be represented by the following formula:
wherein R and R' may be the same or different hydrocarbyl radicals containing from 1 to 18, preferably 2 to 12, carbon atoms and including radicals such as alkyl, alkenyl, aryl, arylalkyl, alkaryl and cycloaliphatic radicals. Particularly preferred as R and R' groups are alkyl groups of 2 to 8 carbon atoms. Thus, the radicals may, for example, be ethyl, n-propyl, i-propyl, n-butyl, i-butyl, sec-butyl, amyl, n-hexyl, i-hexyl, n-octyl, decyl, dodecyl, octadecyl, 2-ethylhexyl, phenyl, butylphenyl, cyclohexyl, methylcyclopentyl, propenyl, butenyl. In order to obtain oil solubility, the total number of carbon atoms (i.e. R and R') in the dithiophosphoric acid will generally be about 5 or greater. The zinc dihydrocarbyl dithiophosphate can therefore comprise zinc dialkyl dithiophosphates. The present invention may be particularly useful when used with lubricant compositions containing phosphorus levels of from about 0.02 to about 0.12 mass %, such as from about 0.03 to about 0.10 mass %, or from about 0.05 to about 0.08 mass %, based on the total mass of the composition. In one preferred embodiment, lubricating oil compositions of the present invention contain zinc dialkyl dithiophosphate derived predominantly (e.g., over 50 mol. %, such as over 60 mol. %) from secondary alcohols.
[0061] Oxidation inhibitors or antioxidants reduce the tendency of mineral oils to deteriorate in service. Oxidative deterioration can be evidenced by sludge in the lubricant, varnish-like deposits on the metal surfaces, and by viscosity growth. Such oxidation inhibitors include hindered phenols, alkaline earth metal salts of alkylphenolthioesters having preferably C5 to C12 alkyl side chains, calcium nonylphenol sulfide, oil soluble phenates and sulfurized phenates, phosphosulfurized or sulfurized hydrocarbons, phosphorous esters, metal thiocarbamates, oil soluble copper compounds as described in U.S. Patent No. 4,867,890, and molybdenum-containing compounds.
[0062] Typical oil soluble aromatic amines having at least two aromatic groups attached directly to one amine nitrogen contain from 6 to 16 carbon atoms. The amines may contain more than two aromatic groups. Compounds having a total of at least three aromatic groups in which two aromatic groups are linked by a covalent bond or by an atom or group (e.g., an oxygen or sulfur atom, or a -CO-, -SO2- or alkylene group) and two are directly attached to one amine nitrogen also considered aromatic amines having at least two aromatic groups attached directly to the nitrogen. The aromatic rings are typically substituted by one or more substituents selected from alkyl, cycloalkyl, alkoxy, aryloxy, acyl, acylamino, hydroxy, and nitro groups.
[0063] Multiple antioxidants are commonly employed in combination. In one preferred embodiment, lubricating oil compositions of the present invention contain from about 0.1 to about 1.2 mass % of aminic antioxidant and from about 0.1 to about 3 mass % of phenolic antioxidant. In another preferred embodiment, lubricating oil compositions of the present invention contain from about 0.1 to about 1.2 mass % of aminic antioxidant, from about 0.1 to about 3 mass % of phenolic antioxidant and a molybdenum compound in an amount providing the lubricating oil composition from about 10 to about 1000 ppm of molybdenum.
[0064] Representative examples of suitable viscosity modifiers are polyisobutylene, copolymers of ethylene and propylene, polymethacrylates, methacrylate copolymers, copolymers of an unsaturated dicarboxylic acid and a vinyl compound, interpolymers of styrene and acrylic esters, and partially hydrogenated copolymers of styrene/ isoprene, styrene/butadiene, and isoprene/butadiene, as well as the partially hydrogenated homopolymers of butadiene and isoprene.
[0065] Friction modifiers and fuel economy agents that are compatible with the other ingredients of the final oil may also be included. Examples of such materials include glyceryl monoesters of higher fatty acids, for example, glyceryl mono-oleate; esters of long chain polycarboxylic acids with diols, for example, the butane diol ester of a dimerized unsaturated fatty acid; oxazoline compounds; and alkoxylated alkyl-substituted mono-amines, diamines and alkyl ether amines, for example, ethoxylated tallow amine and ethoxylated tallow ether amine.
[0066] Other known friction modifiers comprise oil-soluble organo-molybdenum compounds. Such organo-molybdenum friction modifiers also provide antioxidant and antiwear credits to a lubricating oil composition. Examples of such oil soluble organo-molybdenum compounds include dithiocarbamates, dithiophosphates, dithiophosphinates, xanthates, thioxanthates, sulfides, and the like, and mixtures thereof Particularly preferred are molybdenum dithiocarbamates, dialkyldithiophosphates, alkyl xanthates and alkylthioxanthates.
[0067] Additionally, the molybdenum compound may be an acidic molybdenum compound. These compounds will react with a basic nitrogen compound as measured by ASTM test D-664 or D-2896 titration procedure and are typically hexavalent. Included are molybdic acid, ammonium molybdate, sodium molybdate, potassium molybdate, and other alkaline metal molybdates and other molybdenum salts, e.g., hydrogen sodium molybdate, MoOCl4, MoO2Br2, Mo2O3Cl6, molybdenum trioxide or similar acidic molybdenum compounds.
[0068] Among the molybdenum compounds useful in the compositions of this invention are organo-molybdenum compounds of the formulae:
Mo(ROCS2)4
and
Mo(RSCS2)4
wherein R is an organo group selected from the group consisting of alkyl, aryl, aralkyl and alkoxyalkyl, generally of from 1 to 30 carbon atoms, and preferably 2 to 12 carbon atoms and most preferably alkyl of 2 to 12 carbon atoms. Especially preferred are the dialkyldithiocarbamates of molybdenum.
[0069] Another group of organo-molybdenum compounds useful in the lubricating compositions of this invention are trinuclear molybdenum compounds, especially those of the formula Mo3SkLnQz and mixtures thereof wherein the L are independently selected ligands having organo groups with a sufficient number of carbon atoms to render the compound soluble or dispersible in the oil, n is from 1 to 4, k varies from 4 through 7, Q is selected from the group of neutral electron donating compounds such as water, amines, alcohols, phosphines, and ethers, and z ranges from 0 to 5 and includes non-stoichiometric values. At least 21 total carbon atoms should be present among all the ligand organo groups, such as at least 25, at least 30, or at least 35 carbon atoms.
[0070] A dispersant - viscosity index improver functions as both a viscosity index improver and as a dispersant. Examples of dispersant - viscosity index improvers include reaction products of amines, for example polyamines, with a hydrocarbyl-substituted mono-or di-carboxylic acid in which the hydrocarbyl substituent comprises a chain of sufficient length to impart viscosity index improving properties to the compounds. In general, the viscosity index improver dispersant may be, for example, a polymer of a C4 to C24 unsaturated ester of vinyl alcohol or a C3 to C10 unsaturated mono-carboxylic acid or a C4 to C10 di-carboxylic acid with an unsaturated nitrogen-containing monomer having 4 to 20 carbon atoms; a polymer of a C2 to C20 olefin with an unsaturated C3 to d10 mono- or di-carboxylic acid neutralized with an amine, hydroxyl amine or an alcohol; or a polymer of ethylene with a C3 to C20 olefin further reacted either by grafting a C4 to C20 unsaturated nitrogen-containing monomer thereon or by grafting an unsaturated acid onto the polymer backbone and then reacting carboxylic acid groups of the grafted acid with an amine, hydroxy amine or alcohol.
[0071] Pour point depressants, otherwise known as lube oil flow improvers (LOFI), lower the minimum temperature at which the fluid will flow or can be poured. Such additives are well known. Typical of those additives that improve the low temperature fluidity of the fluid are C8 to C18 dialkyl fumarate/vinyl acetate copolymers, and polymethacrylates. Foam control can be provided by an antifoamant of the polysiloxane type, for example, silicone oil or polydimethyl siloxane.
[0072] Some of the above-mentioned additives can provide a multiplicity of effects; thus for example, a single additive may act as a dispersant-oxidation inhibitor. This approach is well known and need not be further elaborated herein.
[0073] In the present invention it may also be preferable to include an additive which maintains the stability of the viscosity of the blend. Thus, although polar group-containing additives achieve a suitably low viscosity in the pre-blending stage it has been observed that some compositions increase in viscosity when stored for prolonged periods. Additives which are effective in controlling this viscosity increase include the long chain hydrocarbons functionalized by reaction with mono- or dicarboxylic acids or anhydrides which are used in the preparation of the ashless dispersants as hereinbefore disclosed.
[0074] When lubricating compositions contain one or more of the above-mentioned additives, each additive is typically blended into the base oil in an amount that enables the additive to provide its desired function.
[0075] When lubricating compositions contain one or more of the above-mentioned additives, each additive is typically blended into the base oil in an amount that enables the additive to provide its desired function. Representative effect amounts of such additives, when used in crankcase lubricants, are listed below. All the values listed are stated as mass percent active ingredient.
Table II
| ADDITIVE | MASS % (Broad) | MASS % (Preferred) |
| Metal Detergents | 0.1-15 | 0.2 - 9 |
| Corrosion Inhibitor | 0-5 | 0 - 1.5 |
| Metal Dihydrocarbyl Dithiophosphate | 0.1 - 6 | 0.1 - 4 |
| Antioxidant | 0 - 5 | 0.01 - 3 |
| Pour Point Depressant | 0.01 - 5 | 0.01 - 1.5 |
| Antifoaming Agent | 0 - 5 | 0.001 - 0.15 |
| Supplemental Antiwear Agents | 0 - 1.0 | 0 - 0.5 |
| Friction Modifier | 0 - 5 | 0 - 1.5 |
| Viscosity Modifier | 0.01 - 10 | 0.25 - 3 |
| Basestock | Balance | Balance |
[0076] Fully formulated lubricating oil compositions of the present invention preferably have a TBN of at least 8.5, preferably at least 9, such as from about 8.5 to about 13, preferably from about 9 to about 13, and more preferably from about 9 to about 11 mg KOH/g (ASTM D-2896 and/or ASTM D-4739).
[0077] Fully formulated lubricating oil compositions of the present invention preferably have a sulfated ash (SASH) content (ASTM D-874) of about 1.1 mass % or less, preferably about 1.0 mass % or less, more preferably about 0.8 mass % or less.
[0078] Preferably, fully formulated lubricating oil compositions of the present invention derive at least 5 %, preferably at least 10 %, more preferably at least 20 % of the compositional TBN from ashless TBN sources including at least one compound of formula (I). More preferably, fully formulated lubricating oil compositions of the present invention derive at least 5 %, preferably at least 10 %, more preferably at least 20 % of the compositional TBN from at least one compound of formula (I), and less than 25 %, preferably less than 20 %, more preferably less than 15 % of the compositional TBN from ashless TBN sources other than compounds of formula (I), including basic dispersants.
[0079] Fully formulated lubricating oil compositions of the present invention further preferably have a sulfur content of less than about 0.4 mass %, more less than about 0.35 mass % more preferably less than about 0.03 mass %, such as less than about 0.15 mass %. Preferably, the Noack volatility (ASTM D5880) of the fully formulated lubricating oil composition (oil of lubricating viscosity plus all additives and additive diluent) will be no greater than 13, such as no greater than 12, preferably no greater than 10. Fully formulated lubricating oil compositions of the present invention preferably have no greater than 1200 ppm of phosphorus, such as no greater than 1000 ppm of phosphorus, or no greater than 800 ppm of phosphorus.
[0080] It may be desirable, although not essential to prepare one or more additive concentrates comprising additives (concentrates sometimes being referred to as additive packages) whereby several additives can be added simultaneously to the oil to form the lubricating oil composition. A concentrate for the preparation of a lubricating oil composition of the present invention may, for example, contain from about 5 to about 30 mass % of one or more compounds of formula (I); about 10 to about 40 mass % of a nitrogen-containing dispersant; about 2 to about 20 mass % of an aminic antioxidant, a phenolic antioxidant, a molybdenum compound, or a mixture thereof; about 5 to 40 mass % of a detergent; and from about 2 to about 20 mass % of a metal dihydrocarbyl dithiophosphate.
[0081] The final composition may employ from 5 to 25 mass %, preferably 5 to 18 mass %, typically 10 to 15 mass % of the concentrate, the remainder being oil of lubricating viscosity and viscosity modifier.
[0082] All weight (and mass) percents expressed herein (unless otherwise indicated) are based on active ingredient (A.I.) content of the additive, and/or additive-package, exclusive of any associated diluent. However, detergents are conventionally formed in diluent oil, which is not removed from the product, and the TBN of a detergent is conventionally provided for the active detergent in the associated diluent oil. Therefore, weight (and mass) percents, when referring to detergents are (unless otherwise indicated) total weight (or mass) percent of active ingredient and associated diluent oil.
EXAMPLES
(i) Synthesis of 4-ethoxy-N-(4-methylpentan-2-yl)aniline
[0084] A 1 litre, three-necked round-bottomed flask equipped with a thermometer, addition funnel and a mechanical stirrer was charged with 4-ethoxyaniline (20.00g, 143 mmol), sodium triacetoxyborohydride (38.30g, 171 mmol) previously crushed in a mortar, and 475ml of dichloromethane. The addition funnel was charged with 4-methyl-2-pentanone (15.98g, 157mmol) and 25ml of dichloromethane. The ketone was added slowly to the flask via the funnel and the reaction left to proceed overnight. Following reaction, saturated NaHCO3 solution was added via the funnel. The organic phase was then washed again with saturated NaHCO3 solution, again with water and then dried using MgSO4. After filtration and evaporation under vacuum, 31g of a pale yellow oil was obtained. This product was used in the following step (ii).
(ii) Synthesis of 4-ethoxy-N-(2-ethylhexyl)-N-(4-methylpentan-2-yl)aniline
[0085] A 1 litre, three-necked round-bottomed flask equipped with a thermometer, addition funnel and a mechanical stirrer was charged with 4-ethoxy-N-(4-methylpentan-2-yl)aniline prepared in step (i) above (31.00g, 140 mmol), sodium triacetoxyborohydride (37.50g, 168 mmol) previously crushed in a mortar, and 475ml of dichloromethane. The addition funnel was charged with 2-ethylhexanal (20.58g, 154mmol) and 25ml of dichloromethane. The aldehyde was added slowly to the flask via the funnel and the reaction left to proceed overnight. Following reaction, saturated NaHCO3 solution was added via the funnel. The organic phase was then washed again with saturated NaHCO3 solution, again with water and then dried using MgSO4. After filtration and evaporation under vacuum, 71.3g of a dark red oil was obtained. This product was tested below where it is labelled as Synthesis Example 1.
Synthesis Example 2
(iii) Synthesis of 4-ethoxy-N-(butan-2-yl)aniline
[0086] A 2 litre, three-necked round-bottomed flask equipped with a thermometer, addition funnel and a mechanical stirrer was charged with 4-ethoxyaniline (50.00g, 357 mmol), sodium triacetoxyborohydride (104g, 464 mmol) previously crushed in a mortar, and 700ml of dichloromethane. The addition funnel was charged with 2-butanone (31.2g, 429mmol) and 50ml of dichloromethane. The ketone was added slowly to the flask via the funnel and the reaction left to proceed for two days. The reaction was then quenched by adding saturated NaHCO3 solution via the funnel. The organic phase was then washed with saturated NaHCO3 solution, again with water and then dried using MgSO4. After filtration and evaporation under vacuum, 70.4g of a dark red oil was obtained. This product was used in the following step (iii).
(iv) Synthesis of 4-ethoxy-N-(2-ethylhexyl)-N-(butan-2-yl)aniline
[0087] A 2 litre, three-necked round-bottomed flask equipped with a thermometer, addition funnel and a mechanical stirrer was charged with 4-ethoxy-N-(butan-2-yl)aniline prepared in step (iii) above (70.00g, 140 mmol), sodium triacetoxyborohydride (105g, 471 mmol) previously crushed in a mortar, and 750ml of dichloromethane. The addition funnel was charged with 2-ethylhexanal (58.0g, 435mmol) and 50ml of dichloromethane. The aldehyde was added slowly to the flask via the funnel and the reaction left to proceed for two days. The reaction was then quenched by adding saturated NaHCO3 solution via the funnel. The organic phase was then washed with saturated NaHCO3 solution, again with water and then dried using MgSO4. After filtration and evaporation under vacuum, 108.2g of a dark red oil was obtained. This product was tested below where it is labelled as Synthesis Example 2.
Comparative Example - (Synthesis Example 3)
[0088] 450 g of phenetidine (3.28 moles), 1682 g of 2-ethylhexanal (13.1 moles), 45 g of 10% Pd/C and 2L of dry methanol were charged into a Parr reactor. The reactor was pressurized to 10 bar with hydrogen and stirred with heating to 100°C. The reaction was monitored by HPLC to completion. The reactor was then cooled to room temperature and the catalyst was removed by filtration. Distillation of the reaction mixture yielded 800g of product, the structure of which was confirmed by 1H- and 13C-NMR
TBN Performance
[0090] The basicity of a lubricating oil composition can be determined by acid titration. The resulting neutralization number is expressed as total base number, or TBN, and can be measured using various methods. Method ASTM D-4739 (potentiometric hydrochloric acid titration) was used herein. The ASTM D-4739 method is favoured in engine tests and with used oils to measure TBN depletion/retention.
TBN Testing
[0091] A fully formulated lubricating oil composition containing dispersant, a detergent mixture, antioxidant, ZDDP antiwear agent, pour point depressant and viscosity modifier, in base oil was prepared. This lubricating oil composition, which was representative of a commercial crankcase lubricant, was used as a reference lubricant. Fully formulated lubricating oil compositions containing 1.68 mass% of the compounds of Synthesis Example 1 (Oil A) and Synthesis Example 2 (Oil B) were prepared. Also tested were fully formulated lubricating oil compositions containing 1.00 mass % (Oil C) and 2.00 mass % (Oil D) of the comparative compound of Synthesis Example 3. An additional amount of base oil was added to each of the samples to provide comparable total mass. The TBN of each of the resulting samples was determined in accordance with each of ASTM D4739 (in units of mg KOH/g). The results are shown in Table III below.
Table III
| Example | Ref. | Oil A | Oil B | Oil C | Oil D |
| Reference Sample (g) | 95.0 | 47.5 | 47.5 | 47.5 | 47.5 |
| Added Base Oil (g) | 5.0 | 1.66 | 1.66 | 2 | 1.5 |
| Aniline compound (g) | - | 0.84 | 0.84 | 0.5 | 1.0 |
| Total Weight (g) | 100 | 50 | 50 | 50 | 50 |
| TBN by D-4739 | 8.75 | 10.49 | 11.60 | 8.87 | 8.97 |
| ΔTBN against Ref. by D-4739 | - | 1.74 | 2.85 | 0.12 | 0.22 |
[0092] As shown, the compounds of the invention as used in Oil A and Oil B effectively increased the TBN of the lubricating oil composition as measured by ASTM D-4739, without contributing to SASH content. The comparative compound of Synthesis Example 3 having a di-alkyl branching arrangement not according to the present invention gave no significant improvement in TBN. This was so even in the case of Oil D where a greater amount of compound was used (2.00 mass% vs. 1.64 mass%).
Fluoroelastomer seal compatibility testing.
[0093] Oil A and a similar oil containing N,N-di-n-hexyl-4-ethoxyaniline in place of the compound of the invention were tested in the VW-6/AK6 test. This procedure tests compatibility of lubricating oils with Viton fluoroelastomer seal materials. The elongation at break for a reference oil containing neither compound was on average -34.5% compared to a new seal. For Oil A, the average was -51% and for the oil containing N,N-di-n-hexyl-4-ethoxyaniline, -64%. These tests show that the compound of the invention had a much lower impact on the seal material than the analogous compound bearing two liner alkyl chains.
1. A lubricating oil composition comprising a major amount of oil of lubricating viscosity
and a minor amount of one or more compounds of formula (I):
wherein R1, R2, R3, R4 and R5 independently represent alkyl or substituted alkyl having no aryl substituent.
wherein R1, R2, R3, R4 and R5 independently represent alkyl or substituted alkyl having no aryl substituent.
2. A lubricating oil composition according to claim 1 wherein R1 represents methyl or ethyl, preferably methyl.
3. A lubricating oil composition according to claim 1 or claim 2 wherein R3 represents methyl or ethyl, preferably ethyl.
4. A lubricating oil composition according to any preceding claim wherein R5 represents methyl or ethyl, preferably ethyl.
5. A lubricating oil composition according to any preceding claim having a TBN of at
least 6 mg KOH/g, preferably from 6 to 15 mg KOH/g, as measured in accordance with
ASTM D-4739.
6. A lubricating oil composition according to any preceding claim having a sulfated ash
content of no greater than 1.1, preferably no greater than 1.0, more preferably no
greater than 0.8 mass %.
7. A lubricating oil composition according to any preceding claim wherein at least 10
% , preferably at least 15 %, more preferably at least 20 %, of the compositional
TBN, as measured in accordance with ASTM D-4379 is derived from ashless TBN sources
including at least one compound of formula (I).
8. A lubricating oil composition according to any preceding claim having a sulfur content
of less than 0.4 mass, and no greater than 1200 ppm of phosphorus.
9. A concentrate for the preparation of a lubricating oil composition according to any
preceding claim, the concentrate comprising from 2.5 to 30 mass % of one or more compounds
of formula (I) as defined in claim 1; 10 to 40 mass % of a nitrogen-containing dispersant;
2 to 20 mass % of an aminic antioxidant, a phenolic antioxidant, a molybdenum compound,
or a mixture thereof; 5 to 40 mass % of a detergent; and from 2 to 20 mass % of a
metal dihydrocarbyl dithiophosphate.
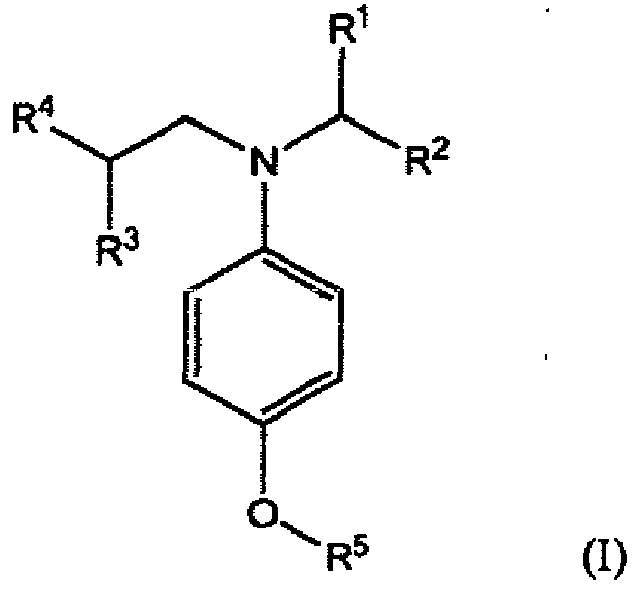
10. A compound of formula (I):
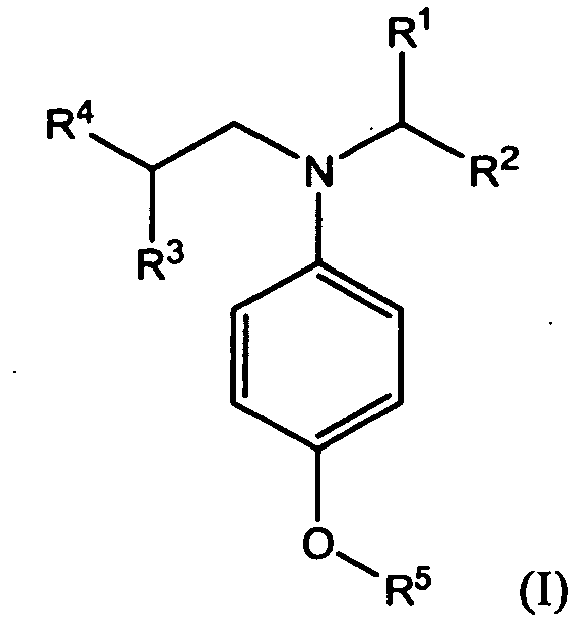
wherein R1, R2, R3, R4 and R5 independently represent alkyl or substituted alkyl having no aryl substituent.
wherein R1, R2, R3, R4 and R5 independently represent alkyl or substituted alkyl having no aryl substituent.
11. A method of increasing the TBN of a lubricating oil composition without concurrently
increasing the sulfated ash content, the method comprising adding to the lubricating
oil composition one or more compounds according to claim 10.
12. The use of a compound according to claim 10 as an ashless lubricating oil composition
TBN source.
1. Schmierölzusammensetzung, die eine größere Menge an Öl mit Schmierviskosität und eine
kleinere Menge einer oder mehrerer Verbindungen der Formel (I):
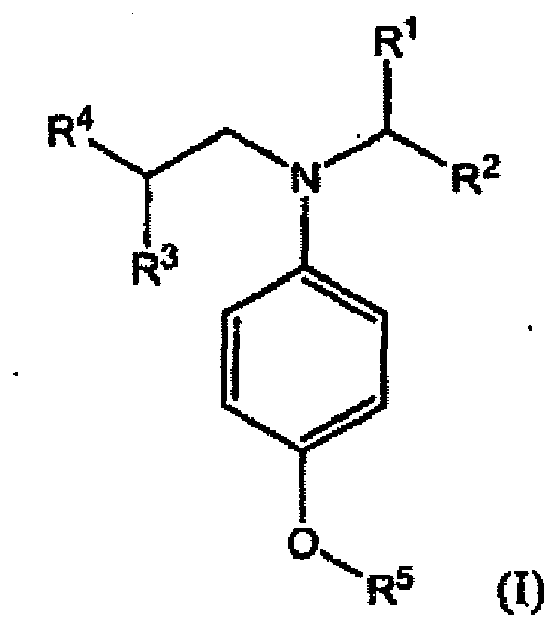
umfasst, in der R1, R2, R3, R4 und R5 unabhängig Alkyl oder substituiertes Alkyl wiedergeben, die keinen Arylsubstituenten aufweisen.
umfasst, in der R1, R2, R3, R4 und R5 unabhängig Alkyl oder substituiertes Alkyl wiedergeben, die keinen Arylsubstituenten aufweisen.
2. Schmierölzusammensetzung nach Anspruch 1, in der R1 Methyl oder Ethyl, vorzugsweise Methyl wiedergibt.
3. Schmierölzusammensetzung nach Anspruch 1 oder Anspruch 2, in der R3 Methyl oder Ethyl wiedergibt, vorzugsweise Ethyl.
4. Schmierölzusammensetzung nach einem der vorhergehenden Ansprüche, in der R5 Methyl oder Ethyl wiedergibt, vorzugsweise Ethyl.
5. Schmierölzusammensetzung nach einem der vorhergehenden Ansprüche, die eine Gesamtbasenzahl
(TBN) von mindestens 6 mg KOH/g, vorzugsweise 6 bis 15 mg KOH/g aufweist, gemessen
mittels ASTM D-4739.
6. Schmierölzusammensetzung gemäß einem der vorhergehenden Ansprüche, die einen Sulfataschegehalt
von nicht größer als 1,1, vorzugsweise nicht größer als 1,0, insbesondere nicht größer
als 0,8 Masse% aufweist.
7. Schmierölzusammensetzung nach einem der vorhergehenden Ansprüche, in der mindestens
10%, vorzugsweise mindestens 15%, insbesondere mindestens 20% der TBN der Zusammensetzung,
wie mittels ASTM D-4379 gemessen, aus aschefreien Quellen von TBN abgeleitet sind,
einschließlich der mindestens einen Verbindung der Formel (1).
8. Schmierölzusammensetzung gemäß einem der vorhergehenden Ansprüche, die einen Schwefelgehalt
von weniger als 0,4 Masse% aufweist und nicht mehr als 1200 ppm Phosphor.
9. Konzentrat zur Herstellung einer Schmierölzusammensetzung gemäß einem der vorhergehenden
Ansprüche, wobei das Konzentrat 2,5 bis 30 Masse% einer oder mehrerer Verbindungen
der Formel I wie in Anspruch 1 definiert, 10 bis 20 Masse% Stickstoff enthaltendes
Dispergiermittel, 2 bis 20 Masse% aminisches Antioxidans, phenolisches Antioxidans,
Molybdänverbindung oder eine Mischung davon, 5 bis 40 Masse% Detergens und von 2 bis
20 Masse% Metalldikohlenwasserstoffdithiophosphat aufweist.
10. Verbindung der Formel (I):
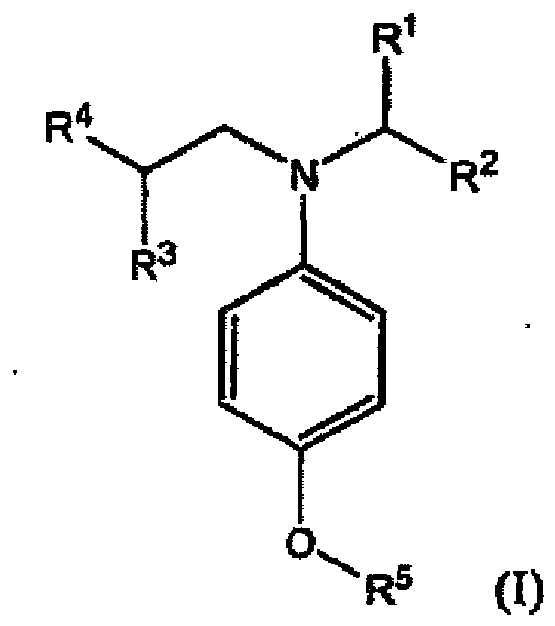
in der R1, R2, R3, R4 und R5 unabhängig Alkyl oder substituiertes Alkyl wiedergeben, die keinen Arylsubstituenten aufweisen.
in der R1, R2, R3, R4 und R5 unabhängig Alkyl oder substituiertes Alkyl wiedergeben, die keinen Arylsubstituenten aufweisen.
11. Verfahren zum Erhöhen der TBN einer Schmierölzusammensetzung ohne gleichzeitige Erhöhung
des Sulfataschegehalts, bei welchem Verfahren der Schmierölzusammensetzung eine oder
mehrere Verbindungen gemäß Anspruch 10 hinzugesetzt werden.
12. Verwendung einer Verbindung gemäß Anspruch 10 als aschefreie TBN-Quelle für eine Schmierölzusamensetzung.
1. Composition d'huile lubrifiante comprenant une quantité dominante d'une huile de viscosité
propre à la lubrification et une petite quantité d'un ou plusieurs composés de formule
(I) :
dans laquelle R1, R2, R3, R4 et R5 représentent indépendamment un groupe alkyle ou alkyle substitué ne comportant aucun substituant aryle.
dans laquelle R1, R2, R3, R4 et R5 représentent indépendamment un groupe alkyle ou alkyle substitué ne comportant aucun substituant aryle.
2. Composition d'huile lubrifiante suivant la revendication 1, dans laquelle R1 représente un groupe méthyle ou éthyle, de préférence méthyle.
3. Composition d'huile lubrifiante suivant la revendication 1 ou la revendication 2,
dans laquelle R3 représente un groupe méthyle ou éthyle, de préférence éthyle.
4. Composition d'huile lubrifiante suivant l'une quelconque des revendications précédentes,
dans laquelle R5 représente un groupe méthyle ou éthyle, de préférence éthyle.
5. Composition d'huile lubrifiante suivant l'une quelconque des revendications précédentes,
ayant un indice de basicité total (TBN) d'au moins 6 mg de KOH/g, de préférence de
6 à 15 mg de KOH/g, tel que mesuré suivant la norme ASTM D-4739.
6. Composition d'huile lubrifiante suivant l'une quelconque des revendications précédentes,
ayant une teneur en cendres sulfatées non supérieure à 1,1, avantageusement non supérieure
à 1,0, plus avantageusement non supérieure à 0,8 % en masse.
7. Composition d'huile lubrifiante suivant l'une quelconque des revendications précédentes,
dans laquelle au moins 10 %, avantageusement au moins 15 %, plus avantageusement au
moins 20 %, du TBN de composition, de la manière mesurée suivant la norme ASTM D-4379,
sont dérivés de sources de TBN sans cendres, comprenant au moins un composé de formule
(I).
8. Composition d'huile lubrifiante suivant l'une quelconque des revendications précédentes,
ayant une teneur en soufre inférieure à 0,4 % en masse et une teneur en phosphore
non supérieure à 1200 ppm.
9. Concentré pour la préparation d'une composition d'huile lubrifiante suivant l'une
quelconque des revendications précédentes, le concentré comprenant 2,5 à 30 % en masse
d'un ou plusieurs composé de formule (I) telle que définie dans la revendication 1
; 10 à 40 % en masse d'un dispersant contenant de l'azote ; 2 à 20 % en masse d'un
antioxydant aminique, d'un antioxydant phénolique, d'un composé de molybdène ou d'un
de leurs mélanges ; 5 à 40 % en masse d'un détergent ; et 2 à 20 % en masse d'un dihydrocarbyl-dithiophosphate
métallique.
10. Composé de formule (I) :
dans laquelle R1, R2, R3, R4 et R5 représentent indépendamment un groupe alkyle ou alkyle substitué ne comportant aucun substituant aryle.
dans laquelle R1, R2, R3, R4 et R5 représentent indépendamment un groupe alkyle ou alkyle substitué ne comportant aucun substituant aryle.
11. Procédé pour augmenter le TBN d'une composition d'huile lubrifiante sans augmenter
conjointement la teneur en cendres sulfatées, le procédé comprenant l'addition à la
composition d'huile lubrifiante d'un ou plusieurs composés suivant la revendication
10.
12. Utilisation d'un composé suivant la revendication 10 comme source de TBN de composition
d'huile lubrifiante sans cendres.
REFERENCES CITED IN THE DESCRIPTION
This list of references cited by the applicant is for the reader's convenience only. It does not form part of the European patent document. Even though great care has been taken in compiling the references, errors or omissions cannot be excluded and the EPO disclaims all liability in this regard.
Patent documents cited in the description
- US5525247A [0007]
- US5672570A [0007]
- US6569818A [0007]
- US20070203031A [0007]
- US4100082A [0008]
- US4200545A [0008]
- US4320021A [0008]
- US4663063A [0008]
- US4708809A [0008]
- SU1825780 [0008]
- US2511750A [0008]
- US3634248A [0008]
- US4269720A [0008]
- US4335006A [0008]
- US4411805A [0008]
- US6242394A [0008]
- US4778654A [0008]
- EP2206764A1 [0009]
- US2045574A [0033]
- US3632511A [0052]
- US3442808A [0053] [0053]
- US3087936A [0054]
- US3254025A [0054]
- US4839071A [0055]
- US4839072A [0055]
- US4579675A [0055]
- US3185704A [0055]
- US4663064A [0055]
- US4612132A [0055]
- US5334321A [0055]
- US5356552A [0055]
- US5716912A [0055]
- US5849676A [0055]
- US5861363A [0055]
- US5328622A [0055]
- US5026495A [0055]
- US5085788A [0055]
- US5259906A [0055]
- US5407591A [0055]
- US4686054A [0055]
- US4867890A [0061]
Non-patent literature cited in the description
- Jn. Org. Chem, 1996, vol. 61, 3489-3862 [0031]
- Engine Oil Licensing and Certification SystemAmerican Petroleum Institute19961200 [0044]
