|
(11) | EP 0 127 064 A1 |
| (12) | EUROPEAN PATENT APPLICATION |
|
|
|
|
|||||||||||||||||||||||||||
| (54) | Rust prevention in processing cans |
| (57) An aqueous solution of a low boiling mildly alkaline amine, such as morpholine, is
applied to drawn and ironed steel, zinc coated ferrous, and tin coated ferrous cans
following cleaning to remove forming lubricant residues and other soil in order to
prevent rust formation on the cleaned cans during subsequent processing. |
BACKGROUND OF THE INVENTION
[0001] This application is a Continuation-In-Part of Patent Application Serial No. 494,746, filed May 16, 1983.
[0002] Metal containers or, more specifically, metal cans used for packaging food and beverages, were formerly manufactured from tinplated steel sheet. In recent years such "tin cans" have been largely replaced by the two piece aluminum can especially for use as beverage containers, e.g. beer and soft drink cans. Such containers can be manufactured at lower cost owing to the malleability of aluminum which allows it to be easily worked to produce thin walled aluminum containers from metal sheet material through a process of drawing and ironing. Additionally the use of aluminum eliminates the need for tin plating since drawn and ironed corrosion-free aluminum cans can be produced by treating the formed and cleaned cans immediately with a corrosion protective conversion coating such as an ALODINE can coating.
[0003] Drawn and ironed cans, or DI cans as they are known in the trade are made by stamping or drawing round flat metal stock into a cuplike intermediate form which is then ironed to produce a conventional can body as a single uniformly thin walled container with integral walls and bottom. For several reasons including the cost of the metal itself, container manufacturers have sought to produce DI cans from steel, particularly-mild steel known as blackplate and while difficulties in forming such cans from steel have been solved the rapidity with which such steel cans corrode or rust remains a problem. The traditional method of tin plating to achieve corrosion resistance does not lend itself to DI cans for the reason that plating after the can is formed is not practical and plating the sheet stock used in forming the cans is not workable since the tin coating does not flow sufficiently during drawing and ironing to provide a continuous tin coating over the entire surface of the DI can. Efforts to eliminate rust formation by using conversion coating treatments immediately after the cans are formed and cleaned have also been thus far unsuccessful owing to the rapiditv with which mild steel rusts, particularly in the event of a line stoppage during manufacture.
[0004] Such treatments, to be useful, must not only prevent rust formation during processing and during the useful life of the container, but must also permit the application of a tightly adherent sanitary lacquer as an interior coating as well as printing a decorative exterior.
[0005] It has now been found that these and other problems in the manufacture of mild steel DI cans, as well as in the manufacture of zinc coated ferrous metal cans and tin coated ferrous metal cans, can be solved by treatment with an aqueous solution of an amine inhibitor in low concentration and having a boiling point sufficiently low to be volatilized during dry off so as to leave no residue which might adversely effect the subsequent coating of the can. Hereinafter when reference is made to DI cans it is intended to also include zinc coated ferrous metal cans and tin coated ferrous metal cans. Accordingly, it is an object of this invention to provide a method of producing rust-free ferrous-based metal containers. More narticularly it ic an obiect nf this invention to produce DI cans from blackplate and to stabilize the formed cans against rust. It is yet a further object of the invention to provide a chemical treatment for maintaining blackplate DI cans free from rust following cleaning of lubricant residues and other soil from the newly formed cans. These and other objects will be more fully understood from the description which follows.
BRIEF SUMMARY OF THE INVENTION
[0006] This invention pertains to an improvement in the method of manufacturing steel and coated ferrous metal cans by a process of drawing and ironing whereby steel sheet stock is drawn and thinned to produce a cuplike cylindrical container of substantially uniform wall thickness. The steel sheet stock used in manufacturing containers by this method as received from the foundry is generally coated with a protective oil such as cottonseed oil or dioctyl sebacate. In preparing the sheet material for drawing and ironing to form containers, lubricants and other forming aids are applied to the surfaces of the sheet. For example, the side which is to become the inside of the container may be coated with a lubricant such as an acrylic wax and the side opposite may be coated with another composition to facilitate uniform metal deformation, such as molybdenum disulfide in an acrylic base. The residues of all of these materials which remain after the drawing and ironing must be thoroughly removed before the containers can be further processed and finished for packing with food or beverage. Generally the drawn and ironed containers are cleaned with aqueous acidic or alkaline cleaning solutions followed by a rinse and drying. It is in this seguence nf treatments that a maier rust problem occurs with blackplate DI cans owing to the fact that the newly cleaned mild steel surfaces are readily oxidized, particularly in the moist environment. In accordance with this invention the newly formed containers can be kept free of rust during subsequent processing by applying to the container surfaces an aqueous solution of an amine corrosion inhibitor in concentrations as low as.100 parts per million or less. The use of an amine inhibitor, such as morpholine, as a corrosion inhibitor for steel is known in the art, for example, Japanese Patent Application JP 81,133,472 discloses a method of protecting mild steel plate from corrosion by coating it with a wax or resin emulsion containing one or more parts of a fatty acid salt of morpholine. Such treatments could not, however, be used in the processing of blackplate DI cans since the emulsion coating would have to be removed before the cans can be further treated with an internal sanitary lacquer and exterior decorative printing or coating. Unlike the prior art application of amine corrosion inhibitors, morpholine or similar amine inhibitors used in accordance with this invention are applied as aqueous solutions in low concentration during or immediately following cleaning and evaporated off during the subsequent drying to leave rust-free clean dry cans which are stable against rusting in further processing; containers treated in this manner are also suitable for application of subsequent protective coatings such as the organic coatings normally applied to provide a suitable foodstuff container.
DETAILED DESCRIPTION OF THE INVENTION
[0007] This invention relates to a method for protecting ferrous-based cans against corrosion by contacting the can surfaces with an aqueous solution of an amine inhibitor. The amines that are useful in this invention are water-miscible amines containing a structural unit of the formula
wherein n is an integer from 0 to 1 and X is O or N. In particular, these water-miscible inhibitory amines are of the formula
wherein R, Rlr R2, R3, R4, R5 and R6 are hydrogen or lower alkyl having from 1 to 4 carbon atoms and may be the same or different, n is an integer from 0 to 1, m is an integer from 1 to 2, X is -0-, -NH-, or -NCH3-, and when n is O and m is 1, R1 and R2 taken together may be -CH2CH2-. The lower alkyl groups may be straight chained or branched and include methyl, ethyl, propyl, isopropyl, n-butyl, iso-butyl, sec- butyl and tert-butyl. Preferably, R3 to R6 are either hydrogen or methyl.
[0008] It is preferred that the amine will remain in solution until substantially all of the water has been evaporated and that any amine remaining on the metal will be sufficiently volatile to evaporate from the metal under the time and temperature conditions of treatment, and further that no decomposition or condensation products of the amine remain on the metal. It is preferred to use rust inhibiting amines of the above formula having boiling points within the range of about 100 to about 210°C, preferably from about 120 to about 170°C. A particularly suitable amine for this purpose is morpholine (R, R3, R4, R5 and R6 are hydrogen, n is 0, m is 1, X is -O-, and R1 and R2 taken together are -CH2CH2-) which is completely miscible in water in all proportions, has a boiling point of 128.9°C and a vapor pressure of 7.0 mm Hg at 20°C.
[0009] Other amines which have been found useful in the practice of this invention include 3-butoxy-propylamine, dimethylamino-2-propanol, dimethylamino-ethanol, diethylamino-ethanol, 2-dimethylamino-2-methyl-1-propanol, 2-amino-2-methyl-1-propanol, monoethanolamine, ethylenediamine, dimethylamino-propylamine, propylenediamine, diethylene-triamine, N-methyl-morpholine, 1,4-piperazine and N,N-dimethyl-1,4-piperazine.
[0010] Mono- and dialkylamines, mono-di-cycloalkyl amines, pyridine, glycols, alkanols, and glycol ethers are unsuitable in the practice of this invention.
[0011] Mild steel plate (blackplate) is drawn and ironed to form blackplate DI cans which are then subjected to a cleaning operation to remove lubricant residues and other soil. The cleaning is generally carried out by passing the cans through a spray washer utilizing a suitable cleaning composition. Any of the acid, alkaline or neutral cleaners known to the art may be employed and the amine inhibitor treatment of this invention works equally well in all cases to prevent subsequent rust formation. Following the cleaning stage which is normally carried out at elevated temperatures, suitably at a temperature between about 120° to about 212°F preferably water to assure a salt-free surface for subsequent coating. The usual process following the rinse is to dry the cans in an oven at temperatures between about 100°C and about 250°C and preferably between about 150° and about 250°C, more suitably at about 200°C, then coat the interior with a sanitary lacquer and the outer surface with a base coat, or vice versa. The outer coating is then inked to apply the desired decorative or descriptive appearance. The major rust problem occurs in the drying stage where the hot moist environment provides near ideal corrosion conditions for the freshly cleaned mild steel surfaces of the blackplate DI cans. At this staqe the cans if properly cleaned are free of any corrosion protection and highly susceptible to any corrosive effects. It might be expected that in the absence of any corrosive agents the cans could be dried and kept in an environment free of corrosive agents until the finish coatings are applied. However, in practice it has been found impossible to produce rust-free blackplate cans by such a clean only method. Even in those cases where there is no visible corrosion it has been found that the initial stages of rust formation referred to as subvisible rust is present. However, cans treated with low concentrations of the inhibitory amine in deionized water can be subsequently dried to clean rust-free surfaces with no trace of subvisible rust and no residual morpholine which volatilizes during drying which is generally carried out at a temperature between about 100°C and about 250°C. The amine treatment also protects against rust formation when treated cans are allowed to dry at ambient conditions such as occur during line stoppage. The aqueous amine treatment is generally applied as a spray immediately following the deionized water rinse. Normal sequence is clean - tap water rinse - deionized water rinse - amine solution final rinse. Optionally the separate deionized water rinse can be eliminated if salt is continuously removed from the amine final rinse. Methods of application other than spray may also be used so long as the entire metal surface (interior and exterior) of the can is thoroughly exposed to the amine solution. The aqueous amine solution can be applied to the can surface at room temperature (about 20°C) which is the most practicable, though higher or lower temperatures can also be used. As a practical matter, the temperature of the spray will not exceed the point at which the amine vaporizes, though vapor treatment is also effective. Normally the amine solution spray will be applied at a temperature between about 15°C and about 35°C. The spray compositions used in this invention consist of only the amine inhibitor and deionized water. It is important that no salts be introduced at this stage since the salt residue will remain on the can surface after evaporation of the water and amine, setting up possible corrosion conditions. The amine is preferably used in a concentration of greater than about 50 ppm. Consistent complete protection against rusting has been obtained by wetting the cans with a deionized water solution containing about 100 ppm of a suitable amine such as morpholine. Higher concentrations of the amines, and in the case of morpholine even up to 100%, can be used though as a practical matter the least amount of morpholine or other amine inhibitor will be used, generally below about 10% by weight. Generally, the amount of amine inhibitor will be between about 20 ppm and about 10,000 ppm and preferably between about 100 and about 5,000 ppm. In the case of morpholine, the most preferred inhibitor, the amount used is generally between about 50 ppm and about 10,000 ppm, preferably between about 100 ppm and about 2,000 ppm and more preferably between about 500 ppm and about 1,500 ppm. The pH of the amine solution should be about pH 8.0 or hiqher, preferably 8.5 or higher and generally between about 8.7 and about 13.5, with a pH between about 10.8 and about 13.5 being especially preferred. The amine spray solution when used in accordance with this invention is also helpful in preventing rusting which might otherwise occur in the event of a line stoppage. For this purpose the manner in which the amine inhibitor is applied is less critical so long as it is present on the metal surface. As a practical matter in a treatment line provided with a final amine rinse stage the amine solution can be readily sprinkled onto the cans in an earlier stage whenever line stoppage occurs, thereby avoiding rusting. Alternatively, amine inhibitor at about 100 ppm or more could be included in the normal operation of each stage and thereby safeguard against rust during stoppage. The presence of the amine inhibitor in the clean and rinse stages will not interfere with the operation or effectiveness of these stages. For optimum protection during dry off and after wash without affecting subsequent coatability of the blackplate cans, it is important that the aqueous solution of the amine inhibitor be totally salt-free and preferably a solution of only the amine and deionized water substantially free of all impurities. The length of time the aqueous solution of the amine remains in contact with the metal surface does not appear to be critical so long as the entire surface of the can is wetted by the solution. Generally a few seconds spray time, e.g. about 1 to about 5 seconds, is sufficient. Cans treated in this way will the humidity conditions normally experienced in a can treatment plant. The invention can be more fully understood by the examples which follow. These examples are intended to illustrate the invention only and are not to be construed as limiting the scope or operation of the invention as understood by those skilled in the art.
EXAMPLE 1
[0012] Blackplate DI cans were cleaned with a commercial cleaning solution using RIDOLINE 666, a can cleaner available from Amchem Products, Inc., Ambler, PA, at 150°C for 21 seconds spray. The cans were rinsed in tap water followed by a deionized water rinse with dwell time of 20 seconds between each stage. Thereafter the cans were allowed a 40-second dwell time and then dried in an air oven using forced air at 200°C for 3 minutes. All cans evidenced significant rusting with rust spots on both the inside and outside domes and around the outside edge.
[0013] Using the same treatment conditions as above with 100 ppm of morpholine added to the deionized water final rinse (pH 9.6), all cans were completely free of rust following the oven drying. The resulting cans were clean, bright and shiny over the entire interior and exterior surfaces. The test was repeated 5 times and each time the morpholine treated cans were all bright with no trace of rust.
EXAMPLE 2
[0014] Following the procedure of EXAMPLE 1, cans were cleaned and treated with a 100 ppm solution of morpholine in deionized water as the final rinse before drying in the oven. The oven temperature was 400°F, two cans each were dried for 5 minutes
EXAMPLE 3
[0015] All of the cans prepared in EXAMPLES 1 and 2 were stored in a humidor at 20°C and 100% relative humidity. After 12 hours there was no change in any of the cans. The cans were maintained in the humidor for an additional 24 hours at 28°C and 85% relative humidity. At the end of 24 hours there was no change in the morpholine treated cans but the cans given a final rinse with deionized water only had additional light rusting near the edges. The test was continued for an additional 1 day at 35°C and 85% relative humidity. Only the cans rinsed in deionized water showed more rust at the edge. After 4 days there was still no change in the treated cans.
EXAMPLE 4
[0016] The following sequence of steps was carried out with a series of blackplate DI cans and the degree of rust formation observed after each step on various parts of the cans was indicated. The cleaning was done in a spray washer using RIDOLINE 666 for 30 seconds time at 150°F. Observations were made in each case 15 minutes after completion of the treatment. The readings are as follows: rust (-), no rust +, slight rust ±. The results were as shown in the table below. The treatment sequences utilized were:
(a) clean only,
(b) clean and blow off,
(c) clean - blow off - tap water rinse,
(d) clean - blow off - tap water rinse - blow off,
(e) clean - blow off - tap water rinse - blow off - deionized water rinse,
(f) clean - blow off - tap water rinse
-deionized water rinse - blow off,
(g) clean - blow off - tap water rinse - blow off - deionized water rinse - blow off - morpholine final rinse (100 ppm, pH 8.9), and
(h) clean - blow off - tap water rinse - blow off - deionized water rinse - blow off
- morpholine final rinse (100 ppm, pH 8.9) - blow off.
EXAMPLE 5
[0017] Following the procedure of EXAMPLE 2 a number of amines were used in concentrations of 1,000 ppm in deionized water. No rust or residue formation occurred during the oven treatment with any of the amines. The amines were morpholine, 3-butoxypropylamine, dimethylamino-2-propanol, dimethylamino-ethanol, diethylamino-ethanol, 2-dimethylamino-2-methyl-l-propanol, 2-amino-2-methyl-I-propanol, monoethanolamine, ethylenediamine, dimethylamino-propylamine, propylene-diamine, and diethylene-triamine.
1. A method of preventing rust formation on blackplate DI cans, zinc coated ferrous
cans, and tin coated ferrous cans which comprises applying to the cleaned can surfaces
an aqueous solution of an amine of the formula
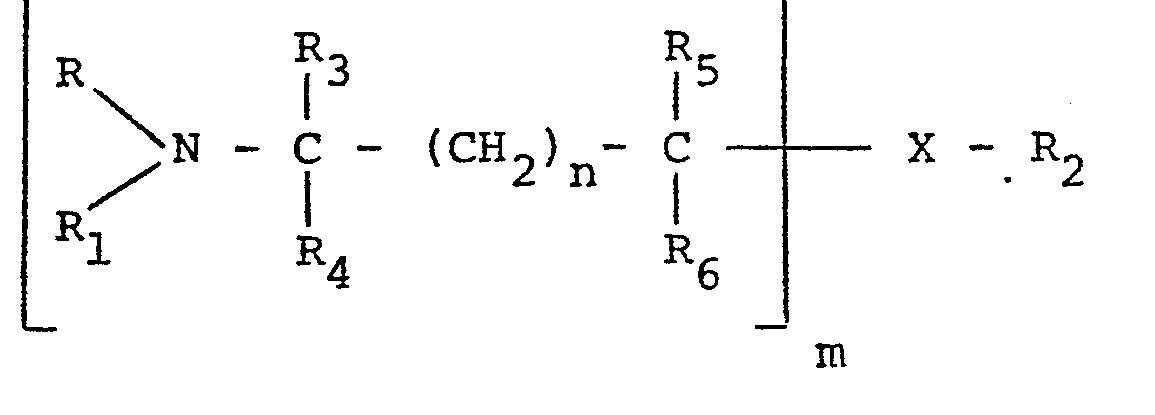
wherein R, R1, R2, R3, R4, R5 and R6 are hydrogen or lower alkyl having from 1 to 4 carbon atoms and may be the same or different, n is an integer from 0 to 1, m is an integer from 1 to 2, X is -0-, -NH-, or -NCH3-, and when n is 0 and m is 1 R1 and R2 taken together may be -CH2CH2-.
wherein R, R1, R2, R3, R4, R5 and R6 are hydrogen or lower alkyl having from 1 to 4 carbon atoms and may be the same or different, n is an integer from 0 to 1, m is an integer from 1 to 2, X is -0-, -NH-, or -NCH3-, and when n is 0 and m is 1 R1 and R2 taken together may be -CH2CH2-.
2. A process according to Claim 1 wherein the amine has a boiling point between about
100 to about 210°C.
3. A process according to Claim 2 wherein the amine has a boiling point between about
120 to about 170°C.
4. The method of Claim 3 wherein the aqueous solution consists of amine inhibitor
and deionized water.
5. The method of Claim 4 wherein the amine inhibitor is morpholine.
6. The method of Claim 4 wherein the morpholine is present in a concentration between
about 50 ppm and about 10,000 ppm and the solution has a pH of about 8.0 and about
13.5.
7. The method of Claim 4 wherein the morpholine is present in a concentration between
about 500 ppm to about 1,500 ppm and the solution has a pH of about 9.2 to about 10.8.
8. A method of preparing rust free blackplate DI, zinc coated ferrous, and tin coated
ferrous cans stable against rustinc under ambient conditions which comprises cleaning
the drawn and ironed cans, and before allowing the cleaned cans to dry, contacting
said cans with an aqueous solution consisting of between about 50 and about 10,000
ppm morpholine in deionized water and thereafter air drying the cans at a temperature
in excess of 100°C.
9. An aqueous spray solution for preventing rust formation when applied to cleaned
blackplate DI, zinc coated ferrous, and tin coated ferrous cans which consists of
morpholine in deionized water.
10. The composition of Claim 9 wherein the concentration of morpholine is between
about 50 ppm and about 10,000 ppm.
